4,4'-Biphenol: A Manufacturer’s Perspective on an Essential Building Block
Historical Development of 4,4'-Biphenol
Through decades in chemical synthesis, certain molecules have shaped entire value chains. 4,4'-Biphenol earned its role as an indispensable intermediate long before composite plastics and advanced resins became ubiquitous. Early patents from the mid-20th century outlined benzene derivatization, and subsequent refinements in oxidative coupling of phenol positioned biphenols for growth in specialty polymers. In our production lines, we witness the legacy of this compound both as a stepping-stone in polymer chemistry and as a refined product whose demand only grows as engineers and chemists push material boundaries.
Product Overview
We produce 4,4'-Biphenol in multi-ton lots, packed to minimize contamination, with batch controls tailored to downstream needs. The production choices stem from years collaborating with polymer makers, pharma developers, and electronics firms. Our experience uncovers specific customer pain points — direct solubility, melting behavior, or trace metal content. Addressing these at the production level shapes not just our workflow, but core product attributes valued in precision applications like high-performance polyarylethers or specialty adhesives.
Physical and Chemical Properties
Handling 4,4'-Biphenol daily, we pay attention to properties beyond tables and datasheets. Its pure white crystalline form indicates batch quality long before testing starts. This compound’s melting point, often cited near 276°C, stands as a sentinel for purity — we see off-spec footage in the melt, with color shifts and residues betraying contamination. High-density packaging keeps this sensitive solid dry: ambient moisture induces caking and spoils the uniformity essential for automated dosing. High solubility in hot organic solvents, paired with low solubility in cold water, informs both storage protocols and every batch’s handling process.
Technical Specifications & Labeling
Specifications matter in every drum we ship. Beyond assay levels, customers scrutinize color, melting range, and specific trace impurities. Rigorous analytical chemistry, using HPLC, GC-MS, and Karl Fischer titration, has become integral — not just for regulatory checks, but for securing process reliability in downstream syntheses. Our labeling documents traceability, batch history, and hazard information laid out by OSHA and REACH. Detailed technical paperwork smooths regulatory compliance for our partners. We invest in glass fiber-reinforced packaging, minimizing residual loss and exposure risks.
Preparation Methods
Industrial production of 4,4'-Biphenol often traces back to oxidative coupling. Using oxidants such as copper(II) salts and catalysts like pyridine, we avoid side reactions and emphasize yield with vigilant temperature control and staged reactant addition in jacketed reactors. Such process experience separates consistently high-purity biphenol from off-grade material. Batch-by-batch optimization for greener practices — for instance, solvent recovery loops and minimized byproducts — ensures regulatory alignment and waste-reduction. Each process tweak reflects hands-on lessons from chemistry, economics, and environmental stewardship.
Chemical Reactions & Modifications
At the bench and in industrial reactors, this biphenolic core tolerates a range of modifications: etherification, esterification, halogenation, and sulfonation all proceed from this reliable skeleton. As a reagent in polymerization, 4,4'-Biphenol’s dual hydroxy moieties serve as reactive anchors for constructing ladder and network architectures. We have supplied tons to polyaryletherketone and liquid crystal polymer projects, and every kilogram delivered reflects nuanced discussions with chemists about byproduct formation, catalyst needs, and downstream purification. Its reactivity profile ranks it among the more versatile phenolic intermediates — careful handling and impurity control during these transformations mean the difference between a robust or brittle polymer.
Synonyms & Product Names
Anyone in manufacturing faces the challenge of nomenclature variation. We document synonyms such as 4,4'-Dihydroxydiphenyl, p,p'-Biphenol, and Biphenol-4,4', and catalog international registry numbers alongside customs data to avoid needless shipment delays. International harmonization remains work in progress. Different industries or regulatory bodies use legacy naming conventions, so every invoice and customs declaration receives detailed synonym mapping. We advise our partners to reference CAS numbers directly, which has proved to reduce confusion in multi-jurisdictional projects.
Safety & Operational Standards
Technical compliance only secures half the risk equation — we train every operator in local and global safety standards, teaching the rapid recognition of overexposure signs and mislabelings, investing in containment infrastructure. 4,4'-Biphenol’s dust should not be inhaled; we enforce dust collection and provide comprehensive PPE guidelines modeled after years of plant incident reports. All storage and transport infrastructure receives grounding and static discharge measures — biphenolic dust can ignite with surprising ease. Incidents in industry often arise from shortcutting established protocols, so our ongoing investment in safety drills, hazard communication, and spill response have measurably reduced downtime and accident rates.
Application Areas
On the factory floor, shipments of 4,4'-Biphenol flow to high-value sectors. Most volumes move into polyarylether and polyarylsulfone resins, critical in electronics insulation, aerospace components, and automotive applications demanding extreme heat resistance. Other customers formulate water-resistant adhesives or optoelectronic layers. We see labs using 4,4'-Biphenol for complex catalyst supports and in the design of semi-rigid dendrimers. Pharmaceutical researchers explore it as a synthetic intermediate in selective estrogen receptor modulators. Importers and end-users push for ever-tighter specifications, driven by innovations that stretch both the molecule’s performance and our production standards.
Research & Development Trends
Internal investments in pilot reactors and analytics enable incremental improvement. Customer-facing projects have tackled reduction in residual metallic contaminants, which matter for electronics and semiconductor applications. Our recent work involved in-situ purification coupled with continuous flow reactors, shaving hours off cycle times and lowering environmental impact. Collaboration brings new ideas: joint ventures with universities have shown biphenol-based frameworks useful as polymer electrolytes. Real-world R&D rarely travels a straight path; feedback loops between our researchers and line workers turn production hiccups into improved batch quality or process shortcuts. Regulatory shifts toward ‘greener’ chemistry prompt us to constantly reevaluate solvent systems, oxidant choices, and energy usage.
Toxicity Research
Our environmental and safety teams monitor ongoing studies on biphenolic toxicity, referencing both in-house animal model data and government registries. Acute oral LD50 values for rats run in the gram-per-kilogram range, but we take lessons from studies showing irritative effects on skin and eyes. Chronic toxicity data remains less conclusive, so every new regulatory report is reviewed by our compliance staff. Engineering controls and exposure limits receive regular review; plant ventilation receives ongoing upgrades, and we design loading and metering systems to keep operators away from local vapor or powder sources. We invite outside auditors for annual reviews, treating toxicity as a shared risk across chemical factories.
Future Prospects
Market surveys point to rising demand for heat-resistant, chemically stable polymers, fueled by electric mobility, telecommunications, and next-generation microelectronics. Researchers probe deeper uses of biphenols as monomers for smart materials — conductive, ion-conducting, or self-healing plastics. We must continue evolving: stricter regulatory regimes mean cleaner synthesis, less waste, and full lifecycle tracing. Circular economy initiatives invite us to consider reclamation of used biphenolic polymers as a resource, not waste. Innovation in separation and purification, as well as the shift towards continuous flow production, promises higher throughput and minimized environmental burden. The factories that thrive in the future will not simply react to demand but actively shape new applications and safer, cleaner processes, putting molecules like 4,4'-Biphenol at the forefront of sustainable chemistry.
At our plant, the production of 4,4'-biphenol forms the backbone of many material innovations. Years of making this compound have shown just how much it supports several industries, especially as trends shift towards advanced polymers and electronics. Our shop floor regularly juggles batches destined for processes as demanding as polyether ether ketone synthesis and as new as next-generation photoresists. Nothing about handling biphenol is generic in practice—it takes well-maintained reactors, robust purification, and a team that knows the hazards and quirks of the chemistry.
The Leap from Lab to Industry
Many think of 4,4'-biphenol as a mere stepping stone chemical, but experience says otherwise. Its influence begins in specialty polymers like PEEK and LCP, where biphenol lays down the basic framework. These polymers show up in enclosures for electronics, pump parts, lightweight gears, and even spacecraft assemblies. The drive to replace metals with high-temperature, corrosion-resistant, and self-lubricating plastics hinges on stable supplies of pure biphenol. Any unexpected dip in consistency or presence of off-spec batches can ripple through supply chains, sometimes putting multi-million dollar contracts at risk. That's pressure you only appreciate after watching a few challenging runs through the purification towers.
Beyond Plastics—Electronics Keep Us on Our Toes
Biphenol's impact doesn’t stop at plastics. The compound sees steady demand from electronics manufacturers building substrates and insulation for high-frequency environments. Having worked with engineers designing circuit boards for telecommunications, I’ve seen how even small impurities in our product can cause defects at scale. The need for controls—especially as circuit patterns shrink—forces our teams to invest in cleaner synthesis routes and tighter analytical controls. It's more than checking a few technical boxes; defect rates get traced straight back to raw materials, with biphenol often under the microscope.
Pharmaceutical Uses and the Analytical Push
Regulations now play a much bigger role as biphenol appears in certain pharmaceutical intermediates and in the preparation of specialty reagents. With updates to regulations across different regions, our teams perform trace analysis on every lot headed to pharma clients. These aren’t just internal requirements—they reflect a decade of stricter oversight from regulators. Here, even isolated impurities must be explained and, if possible, designed out of the process. Achieving that took investment in both process control and staff training. It pays off, though, in trust and repeat business from cautious buyers.
Pain Points and Solutions in Production
Every plant manager knows the struggle of balancing efficiency and purity. We’ve faced shortages of key raw materials, and waste streams that need new treatment technologies to keep up with internal targets and environmental guidelines. The strongest improvements have always come from listening to operators and R&D chemists, not consultants or sales talk. Whether it’s optimizing hydrogenation to avoid by-products or modifying purification to cut down on solvent use, the best ideas rarely surface on the first try. Progress shows up in safer working conditions, less waste, and lower energy bills—there’s no substitute for real-world trial and error.
Facing Tomorrow’s Demands
4,4'-Biphenol finds new uses every year, from liquid crystals in display screens to specialty coatings in harsh environments. As manufacturing partners request more support, technical conversations increasingly revolve around custom purification or tailored blends, not just bulk supply. Our ongoing challenge is to deliver what engineers and scientists need—on time, with traceable quality, and at volumes that match real industry growth. Making biphenol is never routine, and the effort we put in today sets the foundation for tomorrow’s innovations across plastics, electronics, and pharmaceuticals.
The Structure: Insights from a Manufacturer's Perspective
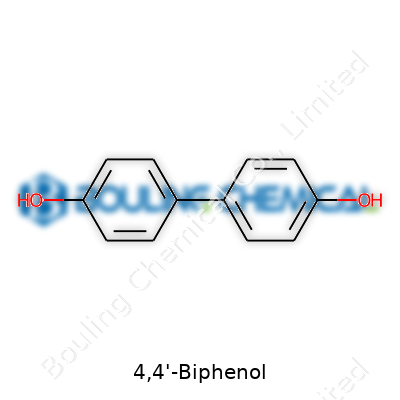
Manufacturing 4,4'-Biphenol puts you in direct touch with a compound that holds both practical value and interesting quirks. In the production hall, this white crystalline solid stands out thanks to its symmetrical structure—two phenolic rings linked through a simple carbon-carbon bond, each marked with a hydroxyl group in the para positions. You get to see what this means up close: those para orientations keep things predictable in processing, with plenty of consistency in purity after crystallization. Physical properties like a melting point around 225°C matter because temperature control affects yield, safety, and the chance of successful downstream operations.
Physical Particulars: Appearance, Handling, and Stability
Unloading 4,4'-Biphenol from the dryer, its solid state and faint odor become familiar. It tends to clump in high humidity, but proper storage curbs this nuisance. The powder disperses easily into reaction vessels, which lets operators avoid dust exposure through station upgrades like local exhaust ventilation. Its low vapor pressure means inhalation hazards stay low during normal operations. In storage, we’ve found that sealed containers and absence of strong oxidizers keep the powder intact for long stretches, avoiding unnecessary waste and extra purification steps.
Chemical Properties Shaped by Manufacturing Realities
The two hydroxyl groups set this material apart. They drive hydrogen bonding and increase water solubility over plain biphenyl. In aqueous or organic solvents, that added polarity defines how it interacts in polymer recipes or as a building block for specialty resins. The same groups hand us conditions for easy esterification or etherification. Consistent reactivity eases integration into high-performance polymers. In our reactors, we watch temperature and pH to avoid unwanted side products, learning more each year about optimizing reaction efficiencies and minimizing solvent volumes.
Safety and Quality: Practical Lessons Learned
Every operator knows 4,4'-Biphenol’s stability but respects its reactive sides. The phenolic groups offer antioxidant properties, which matter whether you’re stabilizing plastics or blocking degradation during sample storage. Under real-world production, we measure incoming raw materials tightly, adjusting purity and moisture preemptively to forestall decomposition and maintain batch-to-batch consistency. Monitoring for trace impurities also prevents troublesome residues in downstream use. Those controls didn’t come from the lab—some rose after troubleshooting sticky plant-wide pumps or an unexpected color change in a finished lot.
Connecting Material to Market Needs
Decades on the floor make one thing clear: how a substance like 4,4'-Biphenol behaves outside the textbook impacts everything from scale-up to application. High melting point improves thermal stability of epoxy matrices, and reliable solubility makes formulation more predictable for coatings and adhesives. If a chemist wants a repeatable outcome, paying attention to real-life chemical and physical quirks becomes crucial. Years of production experience reinforce that careful handling, storage, and in-process monitoring shape not just cost outcomes but the end quality customers expect. Practical knowledge—what actually happens in reactors, bins, and bags—guides the best way forward for synthesizing and supplying dependable 4,4'-Biphenol.
Ground-Level Lessons from the Production Floor
Day in and day out, producing 4,4'-Biphenol calls for sharp attention to detail and disciplined safety habits. We see people talking about “safe handling” all the time, but the specifics matter in a big way when you’re working with a solid organic compound like this. Our teams interact directly with it, so every step counts in keeping operations smooth and safe.
Understanding Real Risks
This chemical doesn’t act dangerously under ordinary temperatures, but exposure can still cause trouble. Dust, for instance, becomes a real irritant. Prolonged or repeated contact with skin and eyes leads to discomfort. We stress the use of adequately fitted goggles and gloves that stand up to aromatic organics. In our experience, nitrile works best. Respiratory protection only comes out when dust becomes unavoidable, such as during grinding, sieving, or loading large batches. Proper ventilation keeps the working environment comfortable and safe.
Preventing Spills and Cross-Contamination
Clean work habits separate the healthy from the hazardous. Staff sweep up any stray powder immediately using non-sparking tools and transfer it to the right waste containers. Vacuum extraction units with HEPA filters help control dust released during packaging. Everybody in our facility knows the golden rule: don’t eat, drink, or store food anywhere near the workstations. This might sound like common sense, but it only takes one lapse for an accident to happen.
Temperature, Humidity, and Safe Storage
Thanks to its melting point above typical room temperatures, 4,4'-Biphenol stays stable under normal warehouse conditions. Long-term quality depends on keeping the area dry. Even trace moisture can cause clumping and affect downstream applications. We use robust packaging — tightly sealed drums or high-density polyethylene bags. Each batch sits well-marked and segregated from acids and strong oxidizers, since these reactivate the material and create unnecessary risks. These segregation rules never get bent, even when we’re short on space.
Training as the Backbone of Safety
Keeping things safe isn’t just about protective gear or proper containers. The real difference comes from frontline staff having a clear understanding of every step involved. We run ongoing training for anyone involved in weighing, transferring, or packing. No employee handles 4,4'-Biphenol unsupervised until they have demonstrated sound technique.
Waste Management and Environmental Responsibility
Spent packaging and off-spec product get dealt with methodically. Waste streams containing traces of the compound head to licensed chemical handlers, instead of being flushed down drains or tossed in with general refuse. We keep exhaustive records on disposal, tracking the movement of every kilo. These steps protect both the environment and the people who live around our operations.
Constant Review and Improvement
Industry standards shift and new research emerges, so our safety protocols don’t stay frozen. We watch regulatory guidance closely and swap methods or products if a safer, proven alternative appears. Staff input gets heard, too. They’re often first to spot opportunities for tighter controls or smarter workflows, and management listens. These small adjustments stack up — creating a work environment where safe handling and storage of 4,4'-Biphenol isn’t a one-off task, but a commitment that runs through the entire operation.
Understanding 4,4'-Biphenol from a Manufacturer’s Bench
Every day on our production floor, chemists work with substances like 4,4'-Biphenol. It looks simple in a bottle—white to off-white crystals, faintly sweet phenolic scent—but behind that, its molecular identity carries real weight for those of us who handle and process it. We engage with its details constantly, and the reason starts right at the molecular scale.
The molecular formula for 4,4'-Biphenol is C12H10O2. Taking a closer look, this reflects two benzene rings joined at the '4' positions of each with one hydroxyl group attached to both. For a working chemist, these numbers aren't trivia. This formula tells you the exact atomic composition, directly affecting material behavior in synthesis and quality control. If the formula slips off by a single hydrogen or oxygen, downstream results miss the mark. That's a headache nobody in process chemistry wants.
Molecular weight adds the next layer. For 4,4'-Biphenol, the value stands at 186.21 g/mol. This number drives the real-world calculations—inventory planning, batching, and safe scale-up. In a plant setting, we calibrate our dosing pumps and mixers based on this figure. If a shipment shows a molecular weight that differs, even in decimal points, red flags go up. We pause everything and question the root cause—are we facing a lot-to-lot variation, contamination, or something else? That determines next steps for both safety and downstream productivity.
Why These Figures Matter Beyond the Textbook
People outside the laboratory see formulas and weights as reference data, boxes to check off. To those of us who form, blend, and purify 4,4'-Biphenol in bulk, these numbers shape much larger decisions. One miscalculation in molecular weight when charging a reactor means lost time, wasted reagents, and unplanned costs. A deviation can introduce impurities and jeopardize entire production runs, especially for clients whose standards leave no room for error. Quality control teams fire up the spectrometers daily—no batch moves forward until the numbers align within strict properties, matching C12H10O2 every single time.
We see errors in published values in online articles or material safety data sheets, some using outdated nomenclature or calculation methods. This confusion risks safety and costs. Teams reading from a flawed source end up with bad math, and that leads to faulty batches. Labeling and downstream analytics catch most discrepancies, but every error costs both time and raw material. It's one reason why we run repeated verifications using industry-trusted databases and instrumentation like NMR or mass spec, checking against in-house reference lots. We have learned that manufacturers bear the burden of accuracy—others down the line rely on our data to perform their own synthesis, safety checks, and regulatory filings.
Addressing Common Pitfalls in Data Management
Maintaining consistency calls for rigorous batch-to-batch traceability. We enforce electronic logs for every lot, linking each entry to precise weight and purity on delivery and after storage. Lab automation limits human error, maintains security, and speeds up recall or audit processes. Where internal systems hit their limits, external partners with ISO-certified labs back us up—double-checking key values for peace of mind.
For anyone scaling up or introducing 4,4'-Biphenol into new process lines, these molecular details should guide every step, not just as facts, but as critical controls for safety, compliance, and efficiency. Real-world manufacturing moves fast, and confidence in numbers like C12H10O2 and 186.21 g/mol means operations keep pace with demand, minimize waste, and keep reputations secure.
A Manufacturer’s Take on Handling 4,4'-Biphenol
Producing 4,4'-Biphenol brings a set of responsibilities for people directly working with the substance, and our crew knows these realities firsthand. The chemical regularly appears in advanced polymer manufacturing and specialty chemical synthesis. For years, our team has worked with 4,4'-Biphenol in large or small batches, so we have seen its behavior in every possible scenario—on hot summer days and in dead-of-winter humidity levels.
Some folks ask if this material is truly hazardous. Chemically speaking, 4,4'-Biphenol brings a health hazard similar to other phenolic compounds. Dust, vapors, or prolonged skin contact can trigger irritation. Overexposure has led to rashes and respiratory complaints among unprotected workers. The substance does not ignite easily, but fine dust can become airborne, and everyone knows dust clouds should never be allowed to hang in the air—one spark, and you risk trouble.
All those years in production have taught us that risk is tied right back to practice. Splash-proof eye protection and fully sealed gloves stand between a safe operator and a painful exposure. Inhalation remains the bigger concern. Even short stints in a non-ventilated space raise the odds of an unpleasant cough. We have relied on well-maintained local exhaust systems and, in more confined situations, half-mask respirators. It’s not about being overcautious; mistakes catch up with you over time. This isn’t a chemical for casual handling.
Strict material handling keeps incidents rare. We store the raw material in designated dry rooms, never leaving opened barrels out in non-production areas. Humidity and cross-contamination only complicate clean-up and disposal. Training for everyone joining the mixing or transfer teams comes before their first shift. That includes real-life drills—our veterans will stop any activity if newcomers forget to check gloves for small tears or walk into controlled zones without a mask.
As a manufacturer, we consult regulatory agencies to shape our standards. Safety Data Sheets from regulatory authorities and detailed occupational limits are always treated as baseline information, not the only resource. Our line supervisors watch for real trends: an uptick in skin complaints or a few folks sneezing more than usual signals a need for review. Emergency showers and eyewash stations sit close to mixing lines because no worker should gamble on "it probably won't happen to me." Even with strict measures, humans make errors, so backup is non-negotiable.
Disposal practices close the safety loop. Unused material never heads to landfill or water streams; instead, it moves under chain-of-custody to high-temperature treatment. If a container has leaked or dust has escaped, cleanup crews use HEPA vacuums and certified PPE, never standard mops or cloths. Routine environmental monitoring keeps surprises away—regular soil, air, and waste samples let us catch any slow-building problems before they turn costly or permanent.
A commitment to safety for 4,4'-Biphenol is not just box-ticking. Experience tells us that everyone—from new recruits to shift leaders—must stay prepared. Putting people first lowers risk, keeps production moving, and upholds the trust our customers and neighbors place in our operation.