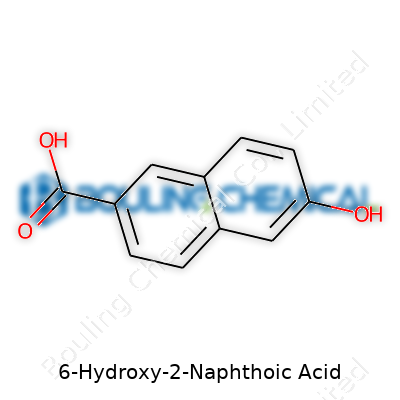
Reflections from the Lab: 6-Hydroxy-2-Naphthoic Acid and Its Chemical Journey
Historical Development
Every chemical has a story. For 6-Hydroxy-2-naphthoic acid, it began in the late 19th century, when aromatic chemistry was transforming industries and academic thinking alike. Early work with naphthalene derivatives opened doors for makers looking to craft new dyes, pigments, and intermediates. Researchers in Europe identified it as a promising candidate during their rush to synthesize new benzene and naphthalene compounds. Phones in our lab may have connectors from recent decades, but glassware remains eerily similar to equipment those pioneers used while melting, oxidizing, and refining these early aromatic compounds. Our recipe has seen improvements: higher-purity starting materials, precision temperature controls, and rigorous quality assurance routines. Yet the soul of the process echoes those experimental days, when each batch felt like new territory.
Product Overview
Our industry values 6-Hydroxy-2-naphthoic acid for its balance of reactivity and stability. Structurally, it’s a two-ring molecule bearing both a hydroxy and a carboxy group, offering two functional “handles” for further chemistry. Production priorities hinge on consistency — yield, purity, and reproducibility. Our process takes raw naphthalene through clean oxidation and precise hydrolysis, followed by extraction and crystallization steps. Standard commercial grades generally exceed 98% purity, supported by HPLC or GC analysis. We supply this product to manufacturers crafting high-performance polymers (like liquid crystal polymers), specialty dyes, and certain pharmaceutical intermediates. We’ve watched its role expand as downstream applications demand better thermal and chemical resistance.
Physical & Chemical Properties
6-Hydroxy-2-naphthoic acid presents as a tan to light-brown solid. Its melting point sits around 225°C, but minor shifts appear batch-to-batch due to water traces or early-stage decarboxylation. It dissolves sparingly in cold water but opens up in hot organic solvents — a boon for downstream reactions. Its phenolic hydroxyl can form strong hydrogen bonds, and the carboxyl group responds to acidic or basic media. Stability comes naturally in sealed containers kept away from light and moisture, but we stress that open handling should to be brief, and ideally under a fume hood.
Technical Specifications & Labeling
We label every drum or bottle not just with batch numbers but also with the key identifiers — CAS number, UN number, and signal words shown in hazard communications. A typical certificate of analysis carries melting range, assay purity, water content by Karl Fischer, and heavy metals by ICP-OES. End-users consistently ask about single impurity profiles and residual solvent traces, so our analytical department invests in updated chromatography libraries and improved detection limits. We take pride in passing regular audits from partners in the electronics and pharma fields.
Preparation Methods
The classic route starts with naphthalene, leading through sulfonation, alkali fusion, and ultimately hydrolysis. Decades ago, sulfonation’s exotherm made this step a nerve-wracking moment, but process improvements have tamed the beast — continuous reactors replace open pots, sensors alert us to runaway conditions. After isolation, recrystallization steps hone purity. Some teams in our industry experiment with oxidative coupling or biocatalytic pathways, searching for milder conditions and greener solvents. Chemical waste from side reactions — mostly inorganic salts and organic tars — receives careful treatment and, where possible, material recovery.
Chemical Reactions & Modifications
This compound plays a starring role as a building block. The carboxylic acid group couples readily to amines or alcohols via esterification or amidation, unlocking access to complex polymers or pharmaceuticals. Photochemical or catalytic hydrogenation of the aromatic ring alters reactivity profiles for some pigment applications. Protecting the phenol group lets chemists steer selectivity towards carboxyl-reactive partners. Over the years, research collaborations with academic partners pushed our pilot team to try new catalysts and solvent strategies, chasing higher yields and sharper selectivity without bumping toxicity or cost.
Synonyms and Product Names
Across the sector, 6-Hydroxy-2-naphthoic acid travels under several names: 6-Hydroxy-beta-naphthoic acid, 2-Carboxy-6-naphthol, and by its registry number, 16712-64-4. Older literature may call it beta-naphthol-2-carboxylic acid or cite proprietary trade names, often linked to colorants or custom syntheses. We stick with clear, harmonized names on every label and technical discussion to avoid mix-ups, as regulatory compliance now hinges on clarity.
Safety & Operational Standards
Direct experience in manufacturing 6-Hydroxy-2-naphthoic acid quickly teaches the importance of real-world safety controls. While this compound isn’t acutely toxic by modern standards, dust inhalation and skin contact risks stand out during bulk handling and drum emptying. Production teams wear nitrile gloves, splash goggles, and certified dust masks. Our protocols draw from GHS, OSHA, and REACH frameworks, as well as lessons learned during near-miss reviews led by operators themselves. We never ignore engineering controls — localized extractor fans, proper drum grounding, and regular slip audits get scheduled as rigorously as product changeovers. Liquid spills on warm stainless can stick and cake, so our cleaning crews use dedicated scrapers and neutralizing rinses. Emergency kits stay close at hand in every work cell.
Application Area
The lion’s share of 6-Hydroxy-2-naphthoic acid flows into performance polymers. Its orthogonal hydroxy and acid groups open doors to polyesters — especially liquid crystal polymers known for their heat and chemical resistance, light weight, and dimensional strength — key for electronics, automotive, and aerospace sectors. Several dye makers depend on this molecule’s scaffold to achieve stability under demanding conditions, translating to brighter and longer-lasting colorants for textiles and printing inks. In drug R&D, medicinal chemists test its naphthalene core for activity in anti-inflammatory and antimicrobial screens. Process innovation at the materials science interface keeps demand lively and pushes our specifications forward each year.
Research & Development
In-house R&D teams experiment with reaction scales and recycling schemes, learning from kilo-lab and pilot-plant runs before committing to larger builds. Instrument upgrades — HPLC, FTIR, NMR — feed back to optimize purification bottlenecks. We also share project data externally, supporting academic or startup teams tackling sustainable synthetic routes or waste minimization. Our technical service chemists field requests from customers who want product modifications: custom particle size, unique polymorphs, or tailored impurity spectra for process validation. We rarely see a year without someone proposing an upgrade or side-project based on fresh literature or patent insights.
Toxicity Research
Safety and environmental profile gets more scrutiny every decade. 6-Hydroxy-2-naphthoic acid doesn’t show acute mammalian toxicity at standard occupational exposures, but chronic tests remain in long-term studies. Our HSE department leans into emerging ecotoxicology data, particularly concerning waterway runoff and soil persistence. We’ve partnered with regulators on fate-and-transport modeling, and adopted best practices for handling process effluent based on up-to-date test outcomes. Any adverse findings get translated into practical workplace changes: routine air and wipe sampling, periodic medical surveillance, and extra engineering controls in high-exposure zones.
Future Prospects
Looking down the line, 6-Hydroxy-2-naphthoic acid’s trajectory will track the evolution of its key end uses, especially advanced polymers and specialty colorants. Process tweaks aimed at greener solvents, higher recycling rates, and integrated continuous production will drive future investments. Academic collaborations and consortium projects already unearth new possibilities — biodegradable copolymers, non-toxic pigment alternatives, and improved analytical markers for trace analysis. Interdisciplinary R&D between chemical engineering, data analysis, and practical bench work brings fresh ideas. As new regulations and customer demands emerge, our responsibility is to refine every part of the production chain — safer chemistry, leaner process flows, and tighter analytical controls, all underpinned by decades of hands-on experience and open dialogue with end users.
Value Built on Experience
Working as a chemical manufacturer, every batch of 6-hydroxy-2-naphthoic acid triggers a chain reaction across different sectors. Customers don't just order this material for the shelf. They usually have a purpose: they use it as a starting point, a foundation for more complex molecules. Our team knows from years on the floor that slight changes in purity or particle size can affect everything down the line, from polymer performance to pigment stability. Crafting consistent lots keeps the wheels turning in several industries that rely on clear, predictable outcomes in their end products.
Building Better Polymers
One major application stands out — polyarylate polymers. These plastics appear in high-performance films, electrical components, and even optical devices. Users search for strength, resistance to heat, and clarity. Every time our operators check for off-color dust or irregular moisture content, they’re making sure downstream partners avoid headaches during extrusion or molding. With markets demanding lightweight, robust materials for electronics and automotive components, the role of 6-hydroxy-2-naphthoic acid grows every year. It’s not just about shipping a drum; it’s about understanding how molecules react during the next step so customers don’t call with a problem after the fact.
Colorants and Dyes: Not Just Synthesizing Pigment
Some customers look to this naphthoic acid as an intermediate for azo dyes and specialty pigments. This branch of chemistry supports textiles, printing inks, and coatings. Fluctuations in shade, solubility, or reactivity can cause recalls or customer complaints. Over decades, we’ve seen how sourcing stable intermediates helps dye manufacturers stay competitive, especially where colorfastness and brightness drive value. A reputation for reliability makes all the difference, especially as regulations around hazardous byproducts grow tighter and clients want fewer worries over final product compliance.
Challenges in Scale-Up and Sustainability
Manufacturing looks simple on paper but every production run brings surprises. Supply chain interruptions, shifting costs of raw chemicals, and environmental regulations affect operations every quarter. Our crew works with chemical engineers side by side to refine processes — controlling waste, reclaiming solvents, and making sure emissions stay within mandated levels. Global customers now ask more about traceability and sustainable sourcing. Persistent effort in waste minimization and resource recovery isn’t just a nod to regulations; it’s about staying competitive as markets shift toward greener products. Methods that let us reuse solvents or recycle byproducts often come from ideas shared on the shop floor, not just in boardrooms.
The Value of Consistency and Collaboration
Production teams, chemists, and customers all want the same outcome: consistent, predictable product that meets strict requirements. Feedback often leads to tweaks in particle control or filtration steps. By acting on those insights, we’ve been able to help partners scale up new polymer grades or stabilize new pigment ranges without drops in yield or rises in cost. This collaboration pays off — not in jargon or generic claims, but in real gains for customers trying to hold their own in tough, competitive markets. That’s the unglamorous but essential value manufacturers bring when delivering chemicals like 6-hydroxy-2-naphthoic acid — reliable building blocks for innovation, quality, and progress.
Crafting Molecules: A Manufacturer’s Perspective
We don’t just read about 6-Hydroxy-2-naphthoic acid in textbooks; we handle it every day, from raw synthesis to final inspection. Its chemical formula, C11H8O3, sounds basic on paper, but every letter and number signifies choices in the plant—vessel materials, temperature control, purification steps. Structurally, this molecule features a naphthalene backbone, fused rings humming with stability, with a carboxylic acid at the second position and a hydroxy group at the sixth. This arrangement steers how we set up reactions, not just for efficiency, but for purity and recovery.
Why Structure Matters to Us and Our Customers
Chemists know a compound’s structure controls its reactivity. Our job doesn’t stop at making a product matching the C11H8O3 formula; we have to control isomerism. If the hydroxy and carboxylic acid groups end up swapped or misplaced, downstream users see it in ruined yields or impure products. A manufacturer’s toughest lesson comes from seeing a zealous student or researcher waste days because a supplier let the 1-hydroxy- or 7-hydroxy isomer slip into the shipment. We don’t take chances on mislabeling or incomplete purification.
Attention to Detail Yields Confidence in Every Batch
In manufacturing, each functional group on this molecule calls for precise process steps. The naphthalene core demands controlled nitration, then selective reduction, then hydrolysis or oxidation at just the right spot. One careless moment spikes impurities, or worse, triggers side reactions that eat yield and complicate downstream filtration. Our teams monitor with real-time spectroscopy to pick up on batch variability before it impacts product. Our customer feedback always points back to one thing: minimal need for rework in their own labs due to our robust in-process controls.
Truth in Transparency: Supporting Industries that Build on Ours
The truth is, we see how much other industries stake on solid intermediates like 6-Hydroxy-2-naphthoic acid. Dyes, polymers, and newer advanced materials use this molecule as a stepping stone to high-performance products. Failures in our supply chain cost time and money right down the line. We don’t chase volume over trust; it helps nobody to send out half-pure batches for the sake of monthly quotas. Years of direct technical dialogue with buyers—sometimes even in-person plant tours—let us learn exactly where our methods fit or fall short in their applications.
Moving the Field Forward
We keep refining our approach year to year, exploring greener solvents and new catalysts that minimize waste and boost yields without sacrificing structure specificity. We invest in training because experienced eyes catch early-stage problems before they lead to recalls or lost trust. Many new employees walk in thinking fine chemicals are all made alike, but a few years in, they grasp the nuances that make the difference between a passable intermediate and a truly reliable 6-Hydroxy-2-naphthoic acid.
What We See in the Plant
6-Hydroxy-2-Naphthoic Acid passes through our hands long before it reaches the chemists, dye houses, or specialty applications. Our crew weighs and transfers hundreds of kilos in a month. From that perspective, safety is more than a checklist—it's a lived responsibility. Each bag we crack open, every filter changeover, each sweep of the floor, reminds us how easy it can be for dust and powders to stray. With this compound, the main worry centers on exposure through both skin and inhalation.
Potential Hazards: Stinging, Dust, and the Air We Breathe
Direct skin contact may cause irritation. Not as dramatic as a caustic burn, but definitely more than a mild itch if powder lingers. Most of us have learned to notice early signs—reddening or dry patches after a day’s work—so gloves and sleeves are always the norm. Eye protection stays on every time a drum gets opened. There’s nothing worse than powder wafting up when scooping from a drum, since fine crystalline solids can reach the eyes or end up inhaled. Even at low concentrations, our crew reports nose or throat irritation if controls slip.
Breathing dust from 6-Hydroxy-2-Naphthoic Acid never ends well in the short term. Without proper extraction or by skipping a mask, coughing and scratchy throats soon follow. Chronic exposure hasn't shown severe cases in our facility, but risk grows when routines slip or new operators ignore instructions.
Beyond the Plant: Fire, Storage, and Shipment Safety
Combustibility doesn't rank high for this acid under normal ambient conditions, but any organic powder deserves respect for dust explosion risk. We enforce regular housekeeping and avoid accumulation on rafters and ledges. Storage remains simple—keep containers sealed and away from strong oxidizers or heat sources. The product keeps well in unopened bags, but bulk storage can present caking if humidity creeps in.
Transportation brings its own story. We load everything under control and documentation, labeling hazards as required. Training our loading team to avoid spills and use spill kits helps prevent the sort of minor releases that might otherwise go unnoticed outside regulated walls.
Handling Mistakes and Emergency Scenarios
Nobody wants to find out safety limits through mistakes. Over the years, we've dealt with small spills and drum mishaps. Quick action—ventilation, PPE, removal to proper disposal—prevents small incidents from escalating. Every crew member knows why MSDS sheets matter; they're neither locked up nor ignored. Working directly with the product, we keep protocols straightforward and practical. Uniforms get washed daily, and open cups or snacks stay outside production zones for a reason.
Improving Safety Attitudes
We teach by repetition: gloves on, mask up, goggles down, sweep up residue. New faces learn by watching veterans who have handled this compound for a decade without incident. Most reported irritation cases have happened when rules weren't followed—a glove torn, goggles worn on the forehead, or hoods not pulled tight during maintenance.
Worksite assessments, air monitoring, refresher training, and easy access to PPE have driven down complaints and fostered a strong culture of mutual correction. We're always exploring better engineering controls: localized extraction, dust suppression systems, or redesigning workstations.
Why Transparency Matters
Our role as the manufacturer brings unique awareness of the practical risks and solutions. By openly discussing actual experience in the plant—not just regulatory talk or hazard phrases—we support downstream users, lab staff, and transport workers alike. Nothing replaces hands-on vigilance and clear communication between all links of the supply chain.
Stability Starts With Consistent Storage
In our experience manufacturing 6-Hydroxy-2-Naphthoic Acid, consistent product quality depends a lot on how the material gets stored. This acid shows impressive stability when kept away from heat and moisture. At our site, we separate it from warehouse areas where temperature can climb above 25°C, since heat can eventually discolor the powder or affect its purity. We reserve dedicated shelving in a shaded, climate-controlled zone—no shortcuts, even for smaller containers or intermediate stocks between production runs.
Moisture Protection: Non-Negotiable
Even trace humidity can encourage caking over time or set off slow hydrolysis reactions that affect downstream applications. We’ve tried both standard poly bags and high-barrier liners, and it’s clear the extra protection makes a difference. Our team seals product right after drying, with fresh inner liners in every drum. From there, sealed packages go directly to the storage area to avoid the swings in ambient air humidity you’ll get in hallways or loading bays on a wet day.
Avoiding Unwanted Chemical Interactions
6-Hydroxy-2-Naphthoic Acid should never sit near substances with strong oxidizing or basic properties. Even small cross-contamination can ruin batches or prompt hazardous reactions. We keep oxidizers, including routine cleaning chemicals, on one side of the warehouse and restrict our acid to its own locked racks. Strict labeling and employee training keep these rules ingrained. Over time, we’ve tightened our protocols after seeing how easy it is for an honest mistake to cause big headaches for quality control.
Using the Right Containers Makes a Difference
Storing this acid in commercial-grade fiber drums lined with polyethylene has stood up well for periods up to several years. Metal containers risk corrosion, and unlined packaging leaves traces of naphthoic odor on the drum walls. After using glass for some test batches, we found it breaks too easily for industrial-scale work. Tested plastic drums resist most wear and offer the best combination of chemical compatibility and low moisture transmission. Every lot receives a tamper-evident seal, and we note inspection dates.
Inventory Rotation and Record-Keeping
Product aging creeps up fast when demand surges turn into slow months. Strict inventory rotation—first produced, first used—has cut down on expired stock. We track batch numbers and manufacture dates in a digital log, triggering reminders for scheduled spot tests. Visual inspections identify off-colors, clumping, or off-spec dust; anything out of order gets quarantined for review. These routine checks head off complaints or failures in the hands of our customers.
Safe Handling Protects Personnel and Product
Beyond storing the material, our crews work with gloves and dust masks, keeping the workspace ventilated and clean. Any spilled acid gets collected with dedicated disposable towels, never swept into shared waste bins. We pour spent packaging into separate disposal streams rather than standard trash, following our commitment to safe and responsible chemical management from shelf to shipping dock.
Lessons Learned on the Floor
After years in production, too many problems trace back to simple corners cut at the storage stage. From unexpected moisture exposure during summer storms to accidental mixing with stronger bases, our best results spring from following the fundamentals every day. Learning these lessons has helped us keep 6-Hydroxy-2-Naphthoic Acid at top quality for everyone relying on our product. Attentive handling, smart storage, and care at every handoff—these steps make chemical manufacturing dependable and safe.
Purity: More Than Just a Number
Running synthesis lines for aromatic compounds day-in and day-out, 6-Hydroxy-2-Naphthoic Acid always stands out in the plant for the checks required. Chemists lean on this intermediate for liquid crystal materials, dyes, pharmaceutical ingredients, and resins that allow for little margin in contamination. High purity keeps downstream reactions predictable. That’s the difference between a batch rerun and a successful, cost-effective output — especially when a customer’s reaction yield depends on limiting organic impurities and metal residues.
After years of manufacturing this molecule, we have seen requests rise for 99% and higher purity lots, particularly from electronics fabricators and research groups. Achieving levels above 99.5% with trace metal control demands close monitoring, right from raw materials to the fine-tuned crystallization and filtration steps. Even during scale-up, managing process consistency means staff run HPLC and GC almost as often as they check reactor pressure. Researchers have described performance issues with even subtle impurities causing color drift or solubility changes in polymers. A direct conversation with a material scientist from a display company convinced us to double-check all possible contaminants, right down to the labeling adhesives used.
Purifying 6-Hydroxy-2-Naphthoic Acid isn’t just about filtration or post-processing—the upstream chemistry must be tight. Excessive thermal exposure during the synthesis leads to hard-to-remove side-products. Many years ago before optimizing this step, we’d see yellowing in certain batches, which taught us the cost of hastily scaling up without proper intermediate analysis.
Packaging: Real-World Supply Practices
As requests from clients have evolved, so has the way we handle the finished acid. Academic labs order a few hundred grams, researchers in specialty chemicals sometimes demand multi-kilo supplies, and larger material companies want drums. Bulk users might want 25 kg fiber drums with liners to keep out atmospheric moisture, since this compound can agglomerate and harden if exposed for long stretches. Routine logistics problems with clumped product taught us early on that robust moisture barriers and desiccant packs keep complaints at bay.
On the smaller end, 100 g to 1 kg glass or HDPE bottles work well for research inventories. Our own team relies on these small packs to avoid wastage or extended exposure. Years ago, standard brown glass seemed sufficient, but over time, customer reports of static buildup with larger plastic bottles led us to trial new anti-static liners for better pouring. Regular feedback loops with end-users—actual bench chemists—showed us that packaging detail impacts everything from measuring ease to long-term performance. Rarely does a week pass without someone calling to discuss a custom size or an inquiry about inert atmosphere packing, especially for sensitive projects.
Clear Communication: The Real Value
Experience has proven effective communication about purity and pack sizes spares both supplier and customer a host of troubles downstream. We see purchasing specialists worry about over-spec’d purity bleeding the budget. On the other hand, a researcher needing ultra-high purity for photonics can risk an entire experiment on a minor batch impurity. Sorting these details early in the conversation has saved us both time and frustration—no surprises, no rejections. Documenting our process controls, impurity profiles, and shelf-life testing isn’t just a regulatory box to tick; it’s how we confirm shipments leave our plant fit for purpose.
It’s easy for technicalities around purity grades and packing formats to fade into jargon, but those variables steer lab results, costs, even process safety. By keeping our technical lines open and production honest, we give our customers the material they need—and we create a smoother workflow for everyone involved.