Anisic Acid: An In-Depth Manufacturer's Perspective
Historical Development Drives Progress in Anisic Acid Manufacture
As a chemical manufacturer with years behind the reactor, stories of anisic acid start with nature herself. Decades ago, plant extracts and essential oils revealed the presence of methylated benzoic acids. Chemists learned early on to isolate p-methoxybenzoic acid from anise seed and star anise, both now valued for their volatile oils and fragrance. The need for broader production and purer material grew with synthetic dyes, flavors, and pharmaceuticals. Early synthetic routes focused on methylation of p-hydroxybenzoic acid or oxidation of p-methoxytoluene. Over the last century, demand for fine chemicals, advances in catalysis, and consumer interests in “nature-identical” ingredients led to both green and scale-friendly paradigms.
Overview: What We Actually Make
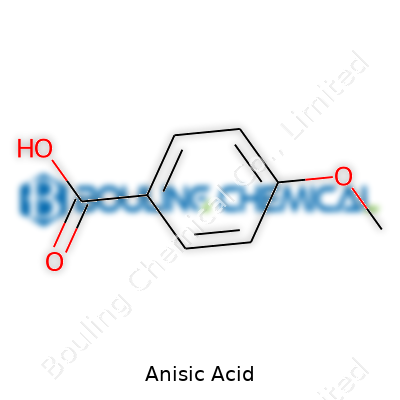
Inside the plant, anisic acid means p-methoxybenzoic acid, a white or off-white crystalline powder. Purity and isomeric content matter more than boastful claims. No shortcuts in purifying, no way around diligent solvent washes, vacuum drying, and the precise temperature control that keeps crystals from discoloring or clumping. Most orders ask for 99% or greater purity, as even 1% of ortho- or meta-methoxybenzoic acid can be a deal-breaker for downstream flavor or pharmaceutical clients. Our labeling reflects international guidelines and uses standard nomenclature, such as 4-methoxybenzoic acid, sometimes echoing the older “draconic acid” from the plant sources.
Physical and Chemical Properties: What Matters in Reality
The melting point centers around 183–185 °C. Melting below 183 °C or an oily residue signals contamination or incomplete crystallization. Water solubility runs low, so we focus on ethanol, ether, and hot organic solvents for process and cleaning steps. Chemically, it stands up to moderate acids and bases, but methoxy substitution on the aromatic ring directs electrophilic attack away from the para position. That makes it attractive for follow-on modifications, like halogenation, amidation, or ester formation. Anisic acid’s moderate acidity (pKa ≈ 4.5) fits applications where benzoic acid is too reactive and p-hydroxybenzoic acid too unstable.
Technical Specifications & Labeling: No Room for Guesswork
Not every customer inspects every drum, but every drum leaving our gate faces batch-level scrutiny: melting point range, IR authentication, NMR purity, heavy metals content, and moisture by Karl Fischer titration. No regulatory latitude exists for unlisted solvents, unapproved batch stabilizers, or skipped sieve analysis. GHS labeling includes hazard codes even if the real-world hazard is mild, because legal and transportation safety demands more than wishful thinking. Harmonized System (HS) codes and REACH status appear on documents. Synonyms used in labeling include anisic acid, p-anisic acid, and 4-methoxybenzoic acid, never deviating from international norms.
Synthesis: Process Details from the Factory Floor
Among manufacturers, the classic two-step approach still rules: methylation followed by oxidation. Starting from phenol, methylation with dimethyl sulfate or methyl iodide gives anisole, which then undergoes oxidation using potassium permanganate or chromic acid to yield the acid. Some lines start with p-cresol, leveraging selective methylation and benzylic oxidation. Recovery and recycling of solvents and reagents drive cost, purity, and environmental load. Monitoring reaction endpoints minimizes byproducts, especially the troublesome ortho- and meta-isomers. Waste stream treatment includes acid neutralization, solvent recovery, and atmospheric scrubbers to stay compliant.
Core Chemical Reactions and Modifications
Our own staff reach for anisic acid when aiming for selective transformations—directed ortho-metalation, formation of acid chlorides for esters and amides, and gentle reduction to anisaldehyde. The methoxy group’s electron-donating effect makes the ring less reactive toward further electrophilic aromatic substitution, offering a degree of predictability rare in benzoic acid derivatives. Derivatization forms the backbone of customized ingredient production, including parabens, flavor esters, and pharmaceutical intermediates. Each reaction must run under controlled atmosphere, as over-oxidation or acid-catalyzed cleavage damages yield and wastes precious raw material.
Applications: Where Anisic Acid Makes an Impact
Fragrance houses value anisic acid’s sweet, mild aroma—think hints of aniseed or fennel—making it a backbone note in perfumes, soaps, and lotions. Pharmaceutical players order tons for intermediate roles in drug synthesis, exploiting its stability and ease of downstream modification. Food and beverage companies use it, within legal limits, as a flavoring component. Recent interest comes from cosmetic preservative manufacturers, where anisic acid acts against yeasts and mold. Specialty resin batches can benefit from its aromatic reactivity, as our partners in advanced polymers can confirm. Pigment and dye chemists find anisic acid indispensable for specific color-fastness and stability traits. Outside these established fields, materials scientists keep exploring its polymerizable derivatives for functional coatings.
Safety and Operational Standards Rooted in Direct Experience
Plants handling any aromatic acid—anisic acid included—prioritize dust control, solvent recovery, and staff training. Early experience with improper ventilation resulted in complaints about nose and throat irritation during filter cake handling and barrel filling, leading to better dust extraction and fitted respirators. Gloves, goggles, and flame-proof workwear reduce risks around open reactions and during transfer of flammable solvents. All routine operations follow ISO-driven procedures for handling, equipment cleaning, and emergency containment. Health impact research lists anisic acid as low-toxicity, but no facility can afford complacency—leaks, spills, or improper storage still lead to occupational exposure. We keep up with international transport classifications, as even benign organics require paperwork and secure drum sealing to avoid cross-contamination.
Research and Development: Constant Pressure to Improve
Process chemists in the lab challenge every old assumption. Higher-yielding methylation routes, safer oxidants, and eco-friendly solvents remain top priorities. We’ve moved away from chromium-based oxidants with clear evidence of Cr(VI) toxicity and regulatory fallout. New catalytic systems reduce waste and require less hazardous material handling. Analytics specialists deploy improved HPLC, chiral GC, and advanced spectrometry to detect trace impurities. Pressure never decreases: buyers want reproducible performance, higher purity, and certified absence of allergens or regulated chemicals. R&D keeps us ahead of regulations and shifts in consumer expectation, whether it’s traceability for “natural” status or limits on byproduct residues.
Toxicity Research and Responsible Disclosure
No reputable manufacturer dismisses worker safety or downstream health effects. Decades of published literature rate anisic acid as low in acute toxicity, though skin or eye exposure always warrants a rinse. Oral toxicity studies show high thresholds compared to simpler benzoic acids. Chronic exposure studies are still sparse, so we push for independent evaluation, especially where new applications touch food or skin contact. Composting and biodegradation studies point to limited persistence and low bioaccumulative risk. Environmental release controls, wastewater filtration, and emissions tracking remain standard.
Looking Forward: Future Prospects for Anisic Acid
Growth shows no sign of stalling. Demand for “nature-identical” aromatics, greener preservatives, and custom building blocks fuels new investments in scalable and sustainable production. Biocatalytic and fermentation routes sit on the table, though cost hurdles remain for large-scale needs. Applications beyond traditional fragrances and pharmaceuticals—functional polymers, advanced anti-corrosive coatings, and specialty personal care—draw attention from new sectors. Regulatory frameworks continue to evolve, especially as markets dig deeper into traceability, environmental footprint, and allergen potential. Chemical manufacturers shoulder the challenge to pair technical excellence with transparency, environmental responsibility, and reliable supply as the world’s expectations keep rising.
A Chemist’s Perspective from Inside the Factory
Watching our drums of anisic acid fill up on the production floor, I see more than just a crystalline powder making its way to the forklift. I see years of applications layered into a single batch. In our line of work, real utility always matters. Let’s talk about where anisic acid finds a solid foothold and why we keep seeing repeat orders for it.
A Real Player in Fragrance and Flavors
Anisic acid, or para-methoxybenzoic acid if you’re reading the label in the lab, stands out in the fragrance and flavors sector. The compound brings out that distinctly sweet, slightly floral tone—people like to call it reminiscent of anise seed or fennel. It’s not just about one note; it rounds off harsh syntheses and smooths heavy formulations in perfumes. In flavor manufacturing, it acts as a backbone for berry, vanilla, or honeybush profiles. Bench chemists notice the stability during cooking or processing. People rarely talk about chemical resilience, but in flavor work, that stability keeps your end product tasting right batch after batch.
Preservation—More than Just a Buzzword
Walk through any personal care production plant and you’ll hear a lot about preservatives. Anisic acid has real-world value here. Its antimicrobial properties cut down on spoilage and play a direct part in extending shelf life. We get steady calls from formulators trying to clean up ingredient decks and reduce synthetic preservative loads. Instead of long, hard-to-pronounce chemicals, they reach for anisic acid as a milder alternative. Though it never replaces a full preservative system, it does allow formulators to pivot away from heavier synthetic agents—a decision based on market pressure and regulatory shifts.
Pharmaceuticals: Small Molecule, Big Potential
In the pharmaceutical world, compounds keep getting scrutinized down to the contaminant level. The raw purity and clear analytical trail that we keep on our anisic acid batches makes a difference. Synthetic pathways use anisic acid as an intermediate for certain active ingredients, especially where a para-methoxy structure is needed. Research teams have experimented with it in anti-inflammatory and antimicrobial formulations, and while not every project leads to a new blockbuster, the ongoing work points to a steady baseline demand.
Everyday Products That Depend on It
Several of our clients in cosmetics add anisic acid to creams, lotions, and cleansers. These formulations benefit from added stability and some extra resistance against bacteria or fungi. The topic of “clean beauty” might feel like a buzzword to outsiders, but in the plant, we see a constant evaluation of what goes in and what gets left out. Raw material suppliers live with that constant audit.
Challenges and Future Steps
Like any specialty chemical, anisic acid comes with a few headaches. Consistency matters. End users notice batch variation and want reliable, high-purity material. We’ve invested in better purification methods—crystallization steps, enhanced filtration, constant tracking on spectroscopy. Regulations keep shifting, especially around allergens in fragrances and allowed preservative blends in personal care. This adds complexity to both production and supply chain.
Labors in chemical manufacturing run deeper than the press releases ever say. Anisic acid’s import goes beyond a single line item on a formula; it shows the fine balance between performance, regulatory demand, and consumer expectations. From bulk orders in tank trucks to precision-packed pails destined for the next perfume trend, each batch tells a story of use, scrutiny, and adaptation.
Our Perspective as a Chemical Manufacturer
Anisic acid lands on ingredient lists for a reason: its antimicrobial properties and light floral aroma give cosmetics formulators a way to protect their products and, sometimes, mask harsher odors. After working with this compound for years, I can say a fair bit about its profile, its handling, and its effect on finished skincare goods. The real story starts in the plant—a lignin breakdown yields anethole, and our process transforms that raw material into anisic acid with precision control over content and purity. That journey shapes every safety consideration along the way.
Safety in cosmetics depends not only on a chemical’s inherent properties but on the purity standards and the concentrations used in finished products. We manufacture anisic acid with a focus on clean, consistent batches because nobody wants to gamble when it lands on skin. Our labs confirm that low concentrations—typically used in creams, lotions, and hair care—fall under recognized safe thresholds. Groups like the Cosmetic Ingredient Review (CIR) Expert Panel and the EU Scientific Committee on Consumer Safety have given their nod after reviewing toxicology results, irritation tests, and patch studies. Acute toxicity numbers for anisic acid rank low, and no links to serious risks like carcinogenicity or reproductive harm have surfaced in publicly available regulatory reports.
Years of direct manufacturing experience have taught us two things: impurities cause more skin reactions than the base compound, and the blend of raw material quality and process control make all the difference. Any manufacturer that skips quality checks courts problems for both consumers and brands. Batch-to-batch consistency matters because microcontaminants—either from side reactions or environmental exposure—can provoke sensitivity in unlucky users. Quality management based on analytical testing like HPLC and GC-MS keeps those risks in check.
The pH of anisic acid lands on the mild side and usually won’t shift a product’s acid balance too far. In the mild concentrations used for preservation or scent, it doesn’t carry the sting of more aggressive acids like salicylic or lactic. Over the years, customers have asked about its allergenic potential. Reports of contact sensitization remain rare, and so far, peer-reviewed data links most issues to product misuse or underground formulas with questionable origins.
Still, trusted cosmetic companies test their entire finished product—not just raw materials—to spot possible adverse effects before launch. We see some brands moving to natural preservation systems and questioning every synthetic. Anisic acid, though it can be plant-derived or chemically synthesized, fills a gap where natural preservation sometimes fails. It offers a broader antimicrobial shield than single-strain extracts. For formulators seeking clean ingredient decks, a well-manufactured anisic acid bridges the space between effective protection and minimal risk.
Any ingredient can trigger issues in rare individuals, and no one expects a zero-risk world. Yet, in thousands of batches shipped to reputable skincare manufacturers, customer safety issues trace back almost entirely to higher-than-listed concentrations or contamination—never the base substance on its own. For end-users and professionals, sticking to products from companies that disclose ingredient sourcing and follow current Good Manufacturing Practices cuts down on surprises.
We’ll keep paying close attention to upstream sourcing and downstream application. Anisic acid, produced in a well-controlled environment and used as intended, earns its place on the modern cosmetic shelf with a strong safety record.
Experience from the Production Floor
From the perspective of an actual chemical manufacturer, anisic acid stands out as more than just a specialty organic acid. Years of hands-on work handling its synthesis, purification, and supply reveal its true value for a host of different industries. This insight comes not from brochures or technical sheets, but from actual production batches, quality checks, and conversations with customers facing real-world challenges.
Versatility Rooted in Chemistry
At its core, anisic acid brings a simple molecular structure: a carboxylic acid with a sweet, aromatic scent. This combination makes it a popular choice for the fragrance and flavor industries. Perfume and food formulators rely on it to deliver a lasting, clean sweet note, not prone to breaking down under normal conditions. As a manufacturer, samples run through a batch can go from a drum in the warehouse to a high-end perfume or food formulation. Brands trust the traceability and purity that come from careful in-house synthesis—something that cannot be guaranteed with imported intermediates, or from uncertain supply chains.
Helping Preserve Quality
Beyond fragrances, anisic acid functions as an effective preservative. Its mild antimicrobial effect extends the shelf life of certain products. With rising consumer concern about overuse of harsh preservatives, companies search for options that work at lower concentrations but do not compromise product safety. Regular quality feedback from customers in personal care and food processing highlights how well anisic acid performs this role. Reliable test results from independent labs, along with our own in-process analysis, continue to demonstrate safety and compliance with global regulatory frameworks.
Enabling Innovation in Pharmaceuticals and Agriculture
Researchers and formulators appreciate anisic acid’s chemical reactivity. By tuning molecules derived from anisic acid, they’ve designed selective herbicides and new drug precursors. Direct feedback from these clients points to anisic acid serving as a chemical building block that reduces total synthesis steps and, in many cases, byproducts are much easier to manage. That matters a lot in chemical manufacturing, where process efficiency and environmental performance drive long-term growth.
Environmental and Supply Chain Considerations
Production methods have evolved to focus on cleaner chemistry, reducing the environmental impact from solvents and reagents. Our own manufacturing line switched to greener oxidation steps several years ago, leading to lower emissions. This helps ease the regulatory burden on both the manufacturer and the downstream customer. Current demand trends point to increasing volumes, but real risk comes not from capacity, but from volatility in supply chains for key raw materials. It pays to source starting materials domestically whenever possible and invest in closed-loop recycling systems for production solvents. This approach keeps pricing stable and supply secure, which buyers appreciate.
Practical Pathways Forward
Anisic acid is not a miracle chemical. It shines in specific settings: as a high-purity ingredient for fragrances, a mild preservative in clean-label foods or cosmetics, and a synthetic building block for advanced chemical synthesis. The best outcomes come from working closely with end users. Tweaks to particle size or purity specs often improve process performance or product appeal, based on real feedback. In the industry, meaningful innovation does not come from marketing alone, but through collaboration and proven reliability over time.
Looking Beyond the Label: What Makes Anisic Acid “Natural” or “Synthetic”?
Conversations around raw materials often circle back to the same basic question: Where does it come from? We hear this a lot about anisic acid. Some buyers seek an all-natural product line for clean-label personal care, flavors, or fine fragrance. Others keep cost and consistency at the forefront. The source and method behind each batch of anisic acid deserve a closer look, because how we define “natural” means a lot more than checking a single box on a form.
Straight from Nature’s Recipe – But with Limits
Anisic acid occurs in certain plants. Star anise and anise seeds both contain compounds that convert into anisic acid under the right conditions. Traditional extraction methods, such as steam distillation followed by purification, pull it directly from botanical matter. We connect with growers, inspect every shipment of source materials, and run small test extractions from time to time. That approach resonates with markets that value a “naturally derived” story. The challenge with fully natural extraction lies in scaling up. Whole crops fluctuate with climate, pests, and farming practices. Extraction yields shift from season to season, which creates bottlenecks when demand surges. For every kilo produced from plants, a lot of land and manual work stands behind it. There are also price swings no one can ignore, which ripple through to final product costs.
Chemical Synthesis: Meeting Market Demands with Precision
The overwhelming majority of anisic acid on the global market is made synthetically. In our factory, we begin with petroleum- or biomass-based feedstocks. These undergo multiple reaction steps—usually oxidation of anisaldehyde or methylation of p-hydroxybenzoic acid—to yield anisic acid with high purity, every batch. Large-scale chemical processes bring quality, affordability, and reliable supply to industries that count on these attributes. Synthetically manufactured anisic acid meets–or surpasses–food, pharma, and fragrance standards, because we control every variable. Analytical testing corroborates molecular structure and purity. Without this route, many customer requests would simply go unfilled.
Regulatory Complications: Navigating “Natural” Claims
Most end-users want transparency, and regulatory agencies take these claims seriously. In Europe, the US, and beyond, authorities define “natural” in different ways. Some certifications require strict traceability from plant harvest to final product bottling. Many flavor and fragrance panels set their own guidelines about what counts as naturally derived—and which production steps or additives are allowed. We keep documentation ready and audit trails clear. That puts extra pressure on both sourcing and quality teams, but clean paperwork builds trust. Synthetic origins often exclude products from earning so-called “natural” claims, which matters especially for buyers crafting position statements for retail or foodservice.
Future Directions: New Pathways and Sustainable Choices
Growing interest in green chemistry challenges us to improve classic synthetic routes. Suppliers now look at biotechnological fermentation using engineered microbes, which take renewable sugars as feedstocks and skip petrochemicals. This method could bridge the gap between full plant extraction and industrial synthesis, offering lower environmental impact and better scalability. As a manufacturer, we follow these advances closely. They carry implications throughout the supply chain. Reliable data from pilot runs and commercial adoption will change how anisic acid’s “nature” is labeled and perceived.
Whether anisic acid comes from a field of star anise or a reactor vessel, the conversation shouldn’t end with a simple label. The methods behind each batch influence safety, traceability, cost, and consistency—everything we value in specialty chemical production. Transparent practices keep customers fully informed and let them chart their own path on the question of natural versus synthetic.
Understanding Real-World Exposure
We have been making anisic acid for decades, shipping it out daily to be used in everything from fragrance formulations to food flavorings. Customers expect quality and reliability, but also peace of mind on safety. The question about allergic reactions comes up often, especially as more brands put ingredient transparency in the spotlight. Our experience producing and handling anisic acid gives us a front-row seat to its effects and questions about safety arise at every customer audit.
What Science and History Tell Us
Anisic acid—known to chemists as p-methoxybenzoic acid—comes from both natural extraction and synthetic production. Its presence in the natural world, found in anise and other plants, means human exposure isn’t new. There are records of this compound dating back more than a century. Over the years, toxicological evaluations, including those from established chemical safety agencies, have not flagged anisic acid as a common allergen. Published literature, incident reports, and our own incident tracking offer no evidence of widespread allergic responses. Even so, manufacturers of flavors and personal care products must look for rare but possible sensitivity cases.
Differences Between Sensitivity and Allergy
We frequently see confusion between irritation and allergic reaction. Pure anisic acid is a mild organic acid—at high concentrations on skin, it can cause local irritation, redness, or slight discomfort, similar to other benzoic acid derivatives. These effects signal sensitivity, not immunological allergy. Allergic responses involve the immune system reacting to the compound as a threat. For anisic acid, the body of scientific and occupational evidence shows that systemic, immune-based reactions are vanishingly rare. In high-volume production settings, reports from our workers rarely mention skin complaints and never true allergies. No regulatory authority classifies it as a skin sensitizer.
Managing Risk Through Process and Knowledge
Even though allergic responses are not a prominent risk, we insist on protective measures in our facility: gloves, ventilation, and handling protocols prevent unnecessary exposure to powders or concentrated solutions. These steps do not only guard against unknown sensitivities—they make the workplace more comfortable. Down the supply chain, formulation chemists dilute anisic acid to parts-per-million levels in consumer products, further reducing the odds of irritation or discomfort. End product surveys reflect this; consumer complaints related to anisic acid are nearly nonexistent compared to other fragrance ingredients.
Why Transparency and Vigilance Remain Essential
The story isn’t just about what the compound does in controlled settings. Sometimes end users raise new questions that did not turn up in the literature or in decades of production. Customers want to know specifics, and so do we. We keep an eye on new studies, safety alerts, and complaint data from large product manufacturers—especially those in food, cosmetics, and fine fragrance. Our technical teams follow developments in allergy science and raw material toxicology, ready to respond or adjust information as needed. If new data emerge suggesting a risk, we are prepared to review our guidance and processes.
Moving Forward Responsibly
Our process as a manufacturer relies on facts, continual monitoring, and a real-world record of usage, not just theoretical risk calculations. By focusing on measured experience, chemical safety guidelines, and responsive communication, we help support brands and consumers who expect their products to be both safe and transparent. Sensitivity reactions remain possible with any substance, but with anisic acid the overwhelming evidence draws the risk line much closer to negligible. Responsible production, honest dialogue, and scientific vigilance remain our tools as the conversation about ingredient safety goes forward.