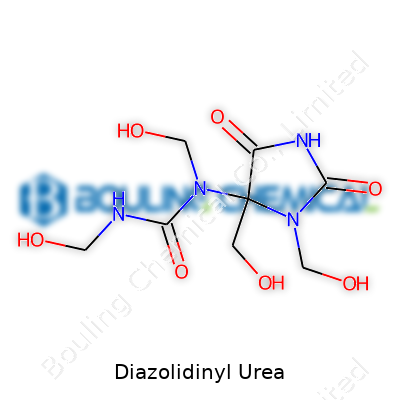
Diazolidinyl Urea: A Perspective from the Chemical Manufacturer
Historical Development
Long before personal care products featured ingredient labels filled with chemical names, Diazolidinyl Urea took its first steps into the cosmetics and industrial landscape. Since the mid-20th century, the rising demand for effective preservatives in aqueous formulations led researchers to consider formaldehyde-donor compounds. The journey from obscure academic notes to widespread commercial use followed the realization that microbial contamination plagued both beauty products and industrial water-based solutions. Manufacturing processes have evolved alongside stricter hygiene standards, resulting in a substance produced under tightly controlled conditions and refined over decades to meet both regulatory pressures and growing consumer scrutiny. We've seen older methods relying on batch mixing transition to more precise, continuous systems, ensuring more consistent product quality. Regulatory discussions in the 1970s and 1980s accelerated investment in analytical infrastructure at production sites, shaping both how manufacturers synthesize and audit Diazolidinyl Urea. Knowledge gained from this development arc guides every plant modification we undertake even today.
Product Overview
Diazolidinyl Urea serves as a broad-spectrum antimicrobial preservative, especially prized in cosmetic creams, shampoos, and lotions, but also relevant in water-based industrial fluids and household cleaners. Our factories formulate it as a white, free-flowing crystalline powder, valued for stable preservation at low effective dosages. Its ability to limit bacterial and fungal growth without notably altering a formula's color or fragrance remains a key driver behind its continued use, especially in products designed for extended shelf life. We consistently monitor its stability under various temperatures and humidity conditions because real-world shelf conditions rarely match laboratory tests. What makes Diazolidinyl Urea distinctly valuable is the balance between preservation strength and compatibility with a range of excipients – something formulators still cite as nontrivial to achieve with alternative agents.
Physical & Chemical Properties
In the manufacturing environment, Diazolidinyl Urea appears as an odorless, white powder with good solubility in water, enabling smooth integration into bulk mixing tanks. Its melting point and hygroscopic character dictate particular storage and handling considerations, such as climate-controlled silos and dry transfer lines. Workers spend time calibrating dosing pumps since the fine particle size allows dusting, requiring dust collection systems and personal protective equipment. It possesses a modest stability window, decomposing gradually when exposed to strong acids, alkalis, or high temperatures over time. Our teams routinely run checks using HPLC and FTIR to catch any hint of decomposition or contamination before bulk release. Given its formaldehyde-release profile, the monitoring of vapor concentrations in enclosed spaces presents a recurring operational challenge, especially during maintenance or cleaning.
Technical Specifications & Labeling
Customers request Diazolidinyl Urea in accordance with standard technical specifications, covering purity, water content, pH range of aqueous solution, and limits for free formaldehyde. As manufacturers, we run each lot against benchmarks laid out in pharmacopeial and industrial references. Quality assurance staff scrutinize labeling to reflect these tests, especially batch numbers, shelf lives, allergen warnings, and safe handling guidelines. There’s a persistent call from global clients for clear labeling regarding formaldehyde release and potential contact sensitivities, pushing us to update documentation in several languages and harmonize hazard pictograms. As labeling expectations grow, so does our investment in barcode traceability technology and digital recordkeeping to guarantee transparency throughout the supply chain. Trace amounts of formaldehyde – even within regulatory limits – still prompt additional dialogue with customers, so up-to-date technical data sheets and safety documentation remain central to our communication process.
Preparation Method
Producing Diazolidinyl Urea involves the reaction of allantoin with formaldehyde and sodium hydroxide or other alkaline agents under carefully controlled temperatures and stirring rates. Our reactors run continuous process monitoring to detect any deviation from target stoichiometry, as variations risk either under-reacted starting material or excessive byproducts. Workers oversee filtration, crystallization, and drying to remove trace impurities and control particle size. Post-synthesis, bulk storage tanks maintain inert atmospheres to inhibit degradation, and rigorous batch sampling occurs before secondary packaging lines. Each step from reactant addition to final drying affects not only overall yield but also the final product’s physical properties. Over years, process engineers refine each aspect to minimize waste and energy use, reflecting broader efforts to reduce resource intensity and operational risk.
Chemical Reactions & Modifications
Most Diazolidinyl Urea in commerce functions as prepared, but in R&D settings, chemists push boundaries by designing analogues with altered release rates or modified antimicrobial spectra. Custom modifications often begin with altering the urea derivative, swapping in alternative amides, or adjusting pH during synthesis to stabilize novel oligomers. Any chemical manipulation, though, triggers a cascade of analytical work to verify antimicrobial activity, decomposition rates, and compatibility with standard cosmetic bases. Our analytical labs focus on quantifying residual formaldehyde and comparing it across modified batches, maximizing safety for end users while maintaining preservation strength. Sometimes, collaborations with downstream users lead us to trial blends with other preservatives to bolster efficacy against particularly resistant microbes. These real-world challenges drive incremental innovation, often based on user feedback looped back to the production floor and laboratory.
Synonyms & Product Names
Diazolidinyl Urea carries an array of alternative names, including Germall II and INCI Diazolidinyl Urea. Technical references and regulatory filings may refer to it as N,N′-Bis(hydroxymethyl)imidazolidin-2,4-dione. Across markets, these variations multiply: our export department regularly aligns product labeling to harmonize terminology across Europe, North America, and Asia, given minor differences in regulatory pathways and tariff codes. Knowledge of synonyms became crucial in avoiding confusion amidst shifting regulatory or procurement standards, making global harmonization a practical necessity well beyond marketing concerns.
Safety & Operational Standards
Our responsibility as a chemical manufacturer begins with worker safety and extends downstream to protect both brand partners and end users. Diazolidinyl Urea, as a formaldehyde-releasing preservative, falls under numerous workplace safety and environmental rules. Production lines feature gas monitors tuned to formaldehyde, local exhaust ventilation, and strict personal protective equipment protocols. Staff receive repeated, scenario-based training covering spill response, process upsets, and skin or eye contact remediation. Plant emergency drills reinforce the need for quick action in case of containment failures. Wastewater treatment facilities run continuously to capture any trace organics or byproducts from washing and floor spills. Downstream, our regulatory affairs teams scrutinize each finished lot against evolving standards for preservative levels in consumer and industrial products, especially in cosmetics. Dependence on accurate technical data means regular third-party audits and membership in industry consortia focused on safer manufacturing.
Application Area
Diazolidinyl Urea found its home mainly in cosmetics: skin creams, facial cleansers, shampoos, and other personal hygiene products. Its biocidal profile also lends itself to cleaners, detergents, and specialty fluids that face microbial spoilage risks. Over the years, formulators come to value it where water activity is high, pH remains mild, and end products require broad compatibility. Customers bring ever-changing needs, sometimes demanding blends tailored for “low free formaldehyde” or for combination with alternative preservatives to tackle new microbial challenges. In industrial settings, process engineers use it to extend useful life of lubricants, coatings, and adhesives exposed to humid environments. Concerns about allergic reactions and regulatory scrutiny push some segments to seek alternatives, but at present, for certain product types and geographies, Diazolidinyl Urea balances cost, performance, and shelf stability beyond what most substitutes achieve.
Research & Development
From the vantage point of a manufacturing R&D lab, the next phase of innovation calls for lower-release analogs, improved stability, and better consumer tolerance. Scientists devote resources to understanding subtle mechanisms of microbial inhibition as regulators question both cumulative exposure and possible allergic sensitivities. Continuing advances in analytical techniques guide reformulation work, while eco-design thinking encourages us to examine not just efficacy but lifecycle impacts – from raw material sourcing through to wastewater treatment after use. Internal R&D teams run side-by-side tests of new preservative systems, benchmarking both raw performance and impact on product aesthetics. Partnerships with universities and contract research groups extend our access to microbiological, toxicological, and consumer data, grounding laboratory findings in the realities of large-scale, long-term use.
Toxicity Research
Our scientists track toxicological data as closely as any process parameter. In-vitro, in-vivo, and clinical patch testing set the bar for consumer-use levels, but reports of contact sensitization, especially among frequent users, spark ongoing scrutiny. Internal reviews continuously check public and proprietary data on DNA interaction, reproductive toxicity, and cumulative formaldehyde exposure. Global regulatory bodies publish varying thresholds for allowable levels, so we tune production and analytical protocols region by region. This means repeated investment in test equipment and regulatory expertise, with emphasis on the latest validated analytical methods for both Diazolidinyl Urea and formaldehyde release. The challenge remains: finding the practical balance between sufficient preservation in high-risk water-based products and the paramount need to minimize user irritation or allergic reactions. We document product-related complaints and adverse event data to inform formulation changes and prioritize R&D.
Future Prospects
Looking to the years ahead, demand for safer, more sustainable preservatives shapes plant upgrades and raw material selection. Green chemistry creeps into process optimization, and our business balances between enabling affordable, effective preservation and meeting new consumer safety requirements. Digital transformation changes everything from batch sampling to real-time monitoring, and regulatory tightening in some regions drives collaborative research into alternatives and hybrid systems. At every turn, the forward path requires attention to safety, efficacy, transparency, and informed application. Those of us producing Diazolidinyl Urea invest not only in production lines but in understanding emerging risks, alternative preservation techniques, and ways to communicate openly with both industrial users and end consumers. The preservative market, like all of specialty chemicals, rewards those who solve technical problems and communicate evidence-backed strategy, rather than relying on legacy formulations or reflexive cost cutting.
Understanding Diazolidinyl Urea’s Place in Manufacturing
In our manufacturing plant, diazolidinyl urea stands out as one of those ingredients that manages to pull more weight than most realize. Every day, batches leave our facility containing this neat little molecule, destined for the shelves of major retailers, independent pharmacies, and clinics worldwide. It’s well known in formulation labs for its ability to help products last, and nowhere is that more important than in industries like cosmetics, skin care, and personal hygiene. If you’ve used a cream or lotion in recent years, chances are good diazolidinyl urea helped keep it free from visible spoilage until you finished it.
Why Manufacturers Trust Diazolidinyl Urea
The pressure to deliver safe, stable products never eases. In our experience, manufacturers select diazolidinyl urea because it works reliably even in complex blends. Every time a formulator chooses an ingredient, they weigh its safety track record, regulatory acceptance, and ability to keep unwanted microbes from taking hold. This preservative consistently checks those boxes. For example, without it or similar molecules, the shelf life of typical personal care products would drop dramatically. Retailers and consumers want as much certainty as possible that products stay good during shipping and storage. That certainty translates into less returned product, fewer customer complaints, and more trust in the brand itself.
Balancing Preservation and Skin Tolerance
Over the years, we see increased attention to what goes into personal products. Consumers search each label, with concerns about synthetic ingredients or allergic reactions. Working directly with bulk buyers and formulators, we’ve tracked complaints and regulatory reports closely. Scientific reviews and market surveillance rarely flag diazolidinyl urea for issues at typical usage levels, though high concentrations never make sense for sensitive skin. Formulators keep levels well within recommended safety margins, and in our own quality control, raw materials always undergo extensive scrutiny. The trick lies in using just enough for safety—without tipping into unnecessary exposure.
The Manufacturing View: Practical and Sustainable Considerations
Production teams need more than just shelf life. Ease of use in automated tanks, clear mixing protocols, and predictable reaction with other ingredients are all serious concerns. Diazolidinyl urea stands out because it dissolves well in water, and its addition doesn’t throw other chemistry out of balance. Scaling up batch sizes, we’ve noted its stability under a wide range of pH and temperature conditions. This resilience is important in large-scale environments where every variable must be tightly managed. Additionally, our regulatory staff monitors safety panels worldwide to anticipate any changes in allowable use—keeping our partners ahead of compliance requirements.
Looking Ahead: Responding to Evolving Expectations
It’s no secret the landscape changes rapidly. Natural alternatives and pressure to “clean up” ingredient lists keep us on our toes. That said, for many products, completely removing synthetics like diazolidinyl urea still presents trade-offs. If reformulation results in shorter shelf life or increased spoilage, supply chains struggle to meet expectations, and waste grows. Our labs keep researching preservation systems, working with partners to minimize exposure while maintaining real-world performance. In the end, our job isn’t just to make chemicals—it’s to help solve real problems, with science as the guide. Diazolidinyl urea, for many years, has done just that.
Understanding the Role in Personal Care
Formulating skincare and cosmetic products that stand up to daily consumer use isn’t a light undertaking. Microbial contamination threatens product stability, shelf life, and even user safety. Diazolidinyl urea enters the picture as a preservative, and we, as direct manufacturers, know its role intimately because we oversee its production at every level—from raw materials sourcing to finished batches heading to formulation labs.
Industry Experience and Safety Evaluation
Diazolidinyl urea has supported the industry’s need for broad-spectrum antimicrobial protection since the 1980s. Decades of batch records and regulatory audits show clear trends: contamination incidents drop when effective preservatives are included and monitored for consistency. For many cosmetic brands, skipping preservation sabotages the shelf life long before the bottle lands on store shelves.
Safety always prompts debate, and rightfully so—no preservative should remain in use without deep scrutiny. Diazolidinyl urea sits on approved preservative lists from regulatory bodies such as the US Food and Drug Administration and the European Commission within concentration limits. Our manufacturing process meets established requirements for purity and formaldehyde release, but we recognize the concerns around irritation and potential allergies.
Direct occupational exposure keeps us vigilant. Bulk handling means strict air and surface monitoring. Reports of skin reactions in end consumers usually trace back to higher concentrations, pre-existing sensitivities, or use in leave-on products that remain for hours against delicate skin. We factor these findings into every batch and keep technical support open for product developers.
The Real-World Challenges
In the real world, the alternative to effective preservation isn’t a clean product—it’s spoiled, microbially compromised goods that could cause far greater harm. Skincare giants and indie formulators alike push for “minimalist” ingredient lists, but a jar of face cream left unprotected can host bacteria, mold, and yeast within days, especially in humid climates. Rigorous testing, not marketing claims, should set the standard.
As preservatives go, diazolidinyl urea doesn’t replace strong cleaning habits or smart packaging. It complements them. Some groups advocate for complete removal because it releases formaldehyde in tiny amounts during its preservative action. We measure those emissions batch by batch, using equipment calibrated against international standards. Finished cosmetic concentrations stay far below thresholds considered to cause harm based on repeated scientific review.
Continuous Improvement and Consumer Communication
Each year, analytical chemists in our plant run stability, preservative efficacy, and irritancy screening as part of our quality commitment. Transparent findings matter—retailers and consumer brands deserve to see data, not just marketing copy. In response to growing demand, our team explores lower-formaldehyde alternatives and combination preservative systems to further reduce risk without sacrificing consumer safety.
Some consumers want “preservative-free” formulas, not fully aware of the risks that come with microbial growth. Our job requires a careful balance: meeting the needs of cosmetic formulators, upholding the trust of end-users, and pushing ongoing innovation behind the scenes. Open dialogue between manufacturers, regulators, brands, and users creates the safest results—not blanket dismissals or shortcuts. As eyes stay on preservatives, so does our commitment to honesty and ongoing safety data.
Understanding Diazolidinyl Urea from a Manufacturer’s Perspective
Diazolidinyl urea often shows up in ingredient lists for cosmetics and personal care products. Those of us making preservatives day in and day out hear the same question now and again—does this ingredient release formaldehyde? That question deserves a clear, experience-based answer instead of evasive language or soft disclaimers.
Production Realities and Chemical Pathways
Our process begins with a reaction between allantoin, formaldehyde, and sodium hydroxide. The purpose is straightforward: make a broad-spectrum antimicrobial that dissolves well in water, holds its ground against bacteria, and extends shelf life without breaking the bank. That’s why so many brands, both big and small, turn to diazolidinyl urea in the first place.
Because the raw materials include formaldehyde, some folks worry about residue or by-products. Chemically, diazolidinyl urea belongs to a family of preservatives known as formaldehyde releasers. Tiny amounts of formaldehyde can come off over time, especially in water-based formulas or under certain heat and pH conditions. Lab analysis backs it up—testing with sensitive methods tracks the free formaldehyde level, and regulation sets clear maximums.
Health Questions and Regulatory Oversight
Formaldehyde draws attention because of its link to sensitivities and its classification as a probable human carcinogen. Understandably, brands want to reassure their customers. As manufacturers working hands-on with this molecule, we keep close watch on regulatory rules across markets. The European Union sets a limit of 0.2% formaldehyde in finished cosmetic products, while US regulations focus on labeling. Each batch of diazolidinyl urea intended for leave-on or rinse-off products gets checked to keep free formaldehyde release within those thresholds.
End-users hardly ever encounter pure diazolidinyl urea. Cosmetic chemists use it sparingly—often between 0.1 and 0.5%—in face creams, lotions, shampoos, and wet wipes. Even at low concentrations, preservatives must do the heavy lifting of keeping bacteria and mold at bay, especially after a jar gets opened and exposed to air and hands.
Alternatives and Solutions
No antimicrobial shield matches the reach of parabens and formaldehyde donors, but concern grows about exposure. Our laboratories and R&D teams see steady demand for new blends. Peptide-based preservatives, organic acids, and essential oil components look promising, but every alternative faces its own hurdles—reduced shelf life, cost spikes, or user reactions.
Switching out diazolidinyl urea often comes with a trade-off. That might mean shorter expiry dates, texture changes, or extra hurdles for microbial safety in challenging formulas. For brands that want to drop formaldehyde releasers altogether, we custom-blend multi-component systems or boost hurdle technology—layering factors like pH adjustment and chelators to build a resilient shield.
Maintaining Transparency
Open disclosure works best. Many of our buyers ask for analysis certificates showing free formaldehyde content or allergen statements up front. As dialogue with consumers sharpens, the bar rises for material traceability. Upgrading traceability systems in the plant helps both us and downstream users maintain trust.
Diazolidinyl urea will likely stay in the rotation so long as it can deliver reliable and broad protection with clear, compliant labeling. As manufacturers, we balance risk with the need for safe products, keeping an eye on ingredient science as it evolves.
What Diazolidinyl Urea Does in Everyday Products
Diazolidinyl urea shows up in a great many personal care products—moisturizers, shampoos, conditioners, even foundations and wet wipes. As a preservative, it helps keep bacteria and mold from ruining water-based creams and lotions. This benefit means products last longer and stay safer for users. From a manufacturer’s perspective, fighting microbial contamination protects both the shelf life and the reputation of finished goods put into the market.
Reports of Irritation and Allergies
Despite the advantages, Diazolidinyl urea attracts its fair share of questions. Some consumers and health advocates raise concerns over skin irritation and allergic reactions. Having supplied this material to dozens of formulators, patterns do emerge. Most people use products with Diazolidinyl urea without any problems. Reports of skin irritation, redness, or rashes remain quite rare when formulations stay within global concentration guidelines. Yet, a small subset of users can experience reactions.
Scientific studies support these observations. Patch testing in dermatological settings confirms that Diazolidinyl urea can act as a mild to moderate allergen, but only in a minority of users. Numbers are similar to reactions from many other common preservatives. Data collected from North America and Europe typically find sensitization rates below 1% in the general population. In our own quality control labs, incidents usually stem from customers with an existing sensitivity to formaldehyde-releasing preservatives. Diazolidinyl urea works by gradually releasing trace amounts of formaldehyde as it performs its antimicrobial job. This aspect deserves attention, as formaldehyde allergies affect a small but significant group of people.
Balance Between Preservation and Sensitivity
Formulators face a real challenge: finding the sweet spot between product safety and minimizing sensitivity risks. Just removing preservatives isn’t practical; the threat from bacteria or molds carries much greater risk, especially in water-rich creams. Instead, manufacturers stick closely to the legal limits set by health authorities like the FDA and European Commission—typically under 0.5% in end products. Rigorous testing ensures safety under normal use conditions.
Sensitive skin is more common than most labels admit. Over years of feedback and trial batches, it’s clear that product design can make a difference. Adding emollients and ingredients that support the skin barrier helps buffer users from possible reactions. Running comprehensive patch tests on volunteers helps catch potential problems before launch. For users with known skin sensitivities, clear ingredient labeling gives people the information they need to avoid triggers.
Moving Forward: Listening to Science and Customers
No preservative solution will ever be perfect for every person. Diazolidinyl urea remains valuable because it guards against contamination without the strong odors or high rates of allergic reactions seen in some alternatives. Risks of irritation and allergy exist, as with any preservative. The available science, careful use in formulation, clear labeling, and open customer feedback all matter in keeping products safe and trustworthy. As manufacturers, taking both safety data and real-world reactions seriously shapes every batch we make.
Understanding Diazolidinyl Urea’s Place in Modern Formulations
Diazolidinyl Urea often appears in ingredient lists for personal care, especially where microbial growth could shorten shelf life. We manufacture this preservative for clients who demand stability in their cosmetic and toiletry products. The industry depends on substances that help keep products safe, but skin compatibility remains a question that cannot be brushed aside, especially for people with delicate or sensitive skin.
Sensitive Skin and Preservative Choices: A Daily Reality
Experiences with raw material sourcing, quality assurance, and production highlight several key points about Diazolidinyl Urea. It releases tiny amounts of formaldehyde over time, something well-known to most formulators. This characteristic extends the life of products, which matters to both manufacturers and consumers, but manufacturers also know that some individuals react to these releasers. People with sensitive skin often report redness, itching, and discomfort in response to common cosmetic preservatives. Dermatologists and safety assessors frequently point out that those with sensitivities may find Diazolidinyl Urea problematic—some studies associate it with increased rates of contact dermatitis, especially among those already showing allergic tendencies.
Long-Term Commitment to Transparency
Across years of production, we have engaged with both formulators and dermatological specialists to push for transparency in labeling and communication. End-users deserve to know what’s in their creams and lotions—especially if they’re prone to issues like eczema or have a history of allergies. Regulatory practices in North America and Europe require explicit labeling of potential allergens. While regulations exist to manage safe usage levels, individual sensitivity does not come with standard thresholds.
Supports and Limitations of Current Preservative Systems
Over the past decade, pressure on manufacturers to drop traditional preservatives has intensified. Customers, retailers, and even major multinational brands have all asked about “formaldehyde-free” options. We developed alternatives—including paraben-free and isothiazolinone-free options—but every system presents its own set of hurdles. Balancing shelf stability with safety for the most reactive skin types tests everyone’s technical know-how. Diazolidinyl Urea answers stability concerns at recommended levels, but for some consumers it’s a source of unwanted reactions.
Future Avenues for Safer Formulations
As ingredient suppliers, we have taken part in many reformulation projects aiming to reduce risk for sensitive skin. Many brands now blend different preservative mechanisms to avoid over-reliance on a single molecule, lowering potential for irritation. More emphasis is placed on challenge-testing and clinical trialing new formulations directly on sensitive panels. Botanical alternatives and newer synthetic molecules are under active development, though more research remains before they can match the performance and cost profile of established systems like Diazolidinyl Urea.
Practical Steps for Manufacturers and End-Users
Pragmatically, the best advice remains to conduct thorough skin compatibility testing before finalizing any formulation for sensitive skin. Ingredient traceability, batch testing, and open dialogue with clients help limit the chance of unpleasant surprises. Sensitive skin cannot always be predicted through paperwork or regulatory compliance. Responsible manufacturers go beyond minimum requirements, tracking feedback and partnering with brands who put consumer well-being first.