Imidazolidinyl Urea: Reflections from the Factory Floor
Historical Roots and Evolution
Imidazolidinyl urea’s journey in industrial chemistry speaks to how modern-day preservation grew from observing natural decay and the limitations that plagued early product shelf lives. In the mid-twentieth century, manufacturing bath and body products would get plagued by persistent spoilage. Even reasonably clean water supplies and careful handling rarely stood up to microbial attack. Our own archives show handwritten batch notes from generations ago, detailing the relentless struggle with short-lived creams and lotions. It didn’t take long for formulators to realize that a single robust preservative could outmatch unreliable blends of acids or alcohols. The appearance of imidazolidinyl urea roughly half a century ago marked a shift in what finished goods could achieve. Shelf lives stretched. Complaints about product spoilage fell. With its entrance, manufacturing changed from fire-fighting to prevention, radically shifting the economics and reliability behind skin care, topical pharmaceuticals, and detergent products.
Overview of the Product
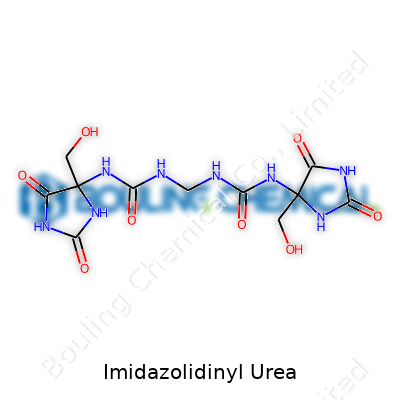
As manufacturers, we recognize imidazolidinyl urea for its reliable role in protecting water-based blends. The molecule hails from the family of formaldehyde-releasing agents. Its ability to hinder the propagation of bacteria broadens the design latitude for personal care and household goods. Unlike simpler preservatives, it does not break down too quickly in finished formulations, which gives manufacturers more confidence, particularly under conditions where product sits unrefrigerated for months. Production batches usually turn out as fine white powders or granular forms, both straightforward to handle in controlled environments. We find these attributes essential for bulk handling—a simple material that accepts blending and dispersal with little fuss.
Physical and Chemical Properties
Imidazolidinyl urea appears as an odorless, white crystalline powder, easy to weigh and introduce into water at room temperature. The compound dissolves easily under most manufacturing conditions, with no visible residue, an advantage during scale-up and reprocessing cycles. During temperature cycling and humidity fluctuations, it holds stability, meaning it doesn't degrade even as raw materials move across different storage areas. Chemical stability remains key—products keep their safety margins as they move from our site to wholesalers, then across retail shelves. Reliability in physical properties ensures that every final product receives true and even preservation, and that’s vital for the safety of the end user.
Technical Specifications, Dosing, and Labeling
Each batch must meet tight specifications, not because regulations insist on it, but because the effects of even small departures—on one hand insufficient bacterial control, on the other excessive formaldehyde release—leave no margin for error. We base allowable levels on trials that measure effectiveness against microbes commonly encountered in filled containers, tying dose ranges directly to residual formaldehyde analyses. Ingredient labeling has become a focus in the age of consumer transparency. We track each lot against local and international requirements, including INCI naming and clarity on potential formaldehyde content where needed. Every finished consignment carries accurate labeling; we invest as much in documentation and batch tracking as in the materials themselves.
Preparation Method and Production Challenges
Our typical synthesis begins with a urea derivative, reacting in controlled vessels with formaldehyde under carefully moderated pH and temperature. Small deviations in reaction time or reagent purity ripple through the product’s physical stability and final microbial effectiveness. Recrystallization and drying finish the process, demanding precise oversight: dry too quickly and the powder quality deteriorates, dry too long and energy costs become punitive without benefit. Process scaling demands real watchfulness—heat exchange gets trickier, pressure differences across filters bring blockages, and timer drift can alter batch consistency. Each new production run goes through analytical quality checks for moisture, purity, particle size, and micro-contaminants. The art of bulk production differs substantially from bench-scale work, with margins for error shrinking as volumes grow. Waste captures, solvent recycling, and vessel cleaning all require diligent attention, especially under stricter emission controls seen across many regions.
Chemical Reactions and Modifications
Chemical relatives—such as diazolidinyl urea—share similar origins but display subtle differences in reactivity and release rates. Tweaking the formaldehyde:urea ratio or shifting the pH window results in preservation agents with distinct kinetics, which affect how slowly formaldehyde gets released into products. Each modification changes interaction not just with microbes, but with co-ingredients; for example, products containing high levels of amines or reducing sugars can impact the preservative’s longevity. Our teams have trialed blends where imidazolidinyl urea partners with organic acids or alcohol-based co-preservatives, sometimes bringing about a broader antimicrobial spectrum and sometimes undermining stability. In the end, only shelf trial data and consumer safety profiles justify process complexity.
Synonyms, Regulatory Names, and Brand Labels
Industry recognizes imidazolidinyl urea by synonyms such as Germall 115, biomollurea, and several trade-specific denominations. Labeling standards often dictate use of the International Nomenclature of Cosmetic Ingredients (INCI), which uniformly requires the full name regardless of trade label or supplier lineage. Regional regulatory agencies—across North America, Europe, and Asia—each lay out unique labeling expectations. We track these nomenclature rules closely to maintain global market access; missteps here can stall entire shipments for months or even result in seizures.
Safety, Exposure, and Operational Controls
Worker safety hinges on real controls throughout every phase. Dust inhalation through handling presents the most obvious risk to plant staff. Inhalation risk control starts with dust collection engineering—extraction arms on blenders, air filtration, and strict personal protective equipment requirements. The core concern from a regulatory perspective centers on formaldehyde release potential, both for production staff and eventual end users. Our testing focuses on minimizing airborne concentration and tracking cumulative exposure, always respecting occupational thresholds. Beyond personnel safety, we maintain strict measures for avoiding product cross-contamination: closed transfer routes, isolated weighing rooms, and digital recipe tracking guard against batch mismatches. Release rates in consumer products remain substantial focal points for toxicological review, so we apply rigorous batch-wise analytics, exporting only product lots with thorough documentation.
Uses Across Industries and Practical Outcomes
Preservative demand cuts across lotion bases, creams, face masks, shampoo, hair gels, and household cleansers. Practically every water-rich formula where bacteria or spoilage is possible can benefit from imidazolidinyl urea’s protection. Our formulation chemists have worked with both new and long-established brands, countering microbial spoilage that arises from user-introduced contamination. Where preservatives did not get used, customer complaints of foul odors, color shifts, or visible mold would spike within months. In high-volume lines, such as low-cost shampoos and creams, this compound often determines whether a product stands up in the real world. That hard-won dependability cements its reputation throughout consumer and commercial product lines.
Research, Toxicity, and Practical Limits
Unlike simpler preservatives such as parabens, imidazolidinyl urea’s reputation remains tied to its status as a formaldehyde donor. Consumer safety groups and regulatory panels inspect it closely for long-term health risks and immediate sensitivities. Our own exposure testing, conducted across multiple years, shows rare but measurable cases of contact dermatitis—mostly among individuals prone to cosmetic allergies. Global health agencies review cumulative formaldehyde release and maintain tolerances at just a few parts per million in finished goods. Regulatory shifts come with every significant toxicology report, so ongoing work by manufacturers keeps pace with new findings. Our development labs routinely test the effectiveness against newer bacterial species and develop newer blends to keep within evolving legal limits. Safety always faces competing demands—preserve the product, avoid overexposing users, and limit the operational footprint during our own factory handling.
Looking Ahead: Adaptation and Innovation
Pressure from market demand and evolving science means no chemical manufacturer can rest on legacy formulas. Researchers in our own pilot plants test new reaction pathways that keep preservation levels high while reducing total formaldehyde exposure. In-house innovation pushes for blends where urea-based compounds work alongside peptides or encapsulated antimicrobials. Some experiments explore bio-derived aldehyde sources, hoping to match the consistency and effectiveness traditional chemistries built up over decades. On the regulatory side, dialogue with agencies and public health experts helps steer investments, ensuring that compliance isn’t a last-minute scramble. Every improvement, whether in plant efficiency, in product labeling, or in the granular control of chemical release, aims to keep preservation reliable without sacrificing safety.
Everyday Roles in Cosmetics and Personal Care
We have made Imidazolidinyl Urea for decades. This preservative shows up almost everywhere—shampoos, moisturizers, creams, foundations. Our customers look for it because their products need to stand up to daily use and months in a bathroom, often in warm, damp environments. Without robust preservatives, bottles of lotion could turn sour fast or grow unpleasant mold. Testing and real-world batch trials always support this: microbe growth picks the path of least resistance, and water-based formulas without protection never last long.
Why Go with Imidazolidinyl Urea?
Every formulating chemist faces a struggle between product shelf life, regulatory demands, consumer trends, and costs. Imidazolidinyl Urea gives an effective pushback against bacteria and yeast, extending a product’s usable lifespan. It's a white, fairly odorless powder that disappears into recipes, so scent and texture never shift. Other preservatives can yellow, react, or disrupt fragrance—those from natural sources often fall short on longevity. Our facility has tested substitution after substitution, but this one gets chosen for its balance of safety assessments and performance in a range of pH conditions.
Consumer Safety and Regulatory Reality
A lot of eyes watch this ingredient—regulatory agencies, advocacy groups, multinational brands. Every batch runs under strict control. We keep free formaldehyde levels well below global standards, regularly sharing raw data with partners. Most reactions attributed to this compound trace back to either improper dose or combining several formaldehyde donors in the same formula, not the ingredient itself used at recommended concentrations. Dermatologists see rare sensitivity, but compare that to the risk of infections from inadequately preserved products. We often walk customers through the supporting studies, helping them balance transparency with the need for reliable preservation.
Facing the Shift Toward “Preservative-Free” Claims
We field constant requests for “clean” or “preservative-free” options now. The marketing pressure in skincare and beauty is real. Many inventive replacements show promise in small-scale applications, but few keep microbial counts down month after month, especially in open, repeatedly hand-touched jars or bottles. Our plant has trialed organic acids, peptides, and botanical extracts. Preservative-free items require single-use packaging or additional refrigeration. Costs climb quickly, and so does waste. Reliable synthetic preservatives, with full documentation and batch-to-batch consistency, continue to fill a need that naturals alone rarely meet at scale.
Moving Toward Trust and Better Solutions
We learn the most from long-term partners who share their experiences with product returns or shifting consumer reviews. The landscape changes, but the basics hold true: any preservative needs scrutiny, traceability, and proof it does its job. Imidazolidinyl Urea gives us a tool that works under all these practical pressures. Manufacturers like ours invest in ongoing toxicology data, new production methods that further lower residuals, and next-generation alternatives, aiming to stay ahead of both regulation and consumer trust.
People often ask whether imidazolidinyl urea belongs in products for sensitive skin. Years of manufacturing this preservative and working directly with raw material sourcing, formulation teams, and regulatory bodies have given us a close look at real-world results—good and bad. It’s easy for headlines to spark concern, but genuine understanding takes looking at all the practical details.
Preservatives Play a Real Role
The modern cosmetics industry runs on preservatives. Unpreserved creams, lotions, and cleansers grow bacteria and mold that can do far more harm than almost any synthetic ingredient. Imidazolidinyl urea brings broad-spectrum activity, meaning it covers a wider range of microbes than many alternatives on the shelf. This compound keeps products shelf-stable for much longer and can reduce the chance someone ends up using spoiled cosmetics. It also works well in water-based formulas, which are notoriously prone to contamination. As chemical manufacturers, we see how much cleaner and more reliable properly preserved products stay—even in hot, humid climates or in open containers that get touched daily.
The Question of Sensitivity
People with highly sensitive skin sometimes react to common preservatives, including imidazolidinyl urea. Skin reactions remain uncommon, but skin biology varies. Experience shows that reactions tend to cluster among individuals already prone to allergies or with impaired skin barriers. Published data suggests true allergy is rare, but it happens. This doesn’t mean imidazolidinyl urea classifies as “unsafe”; it means some people need to monitor their reactions more closely. We’ve worked with customers who run small patch tests first and listen to their skin’s response before using new formulas. The smartest manufacturers check their own formulas for unnecessary overuse of strong preservatives—and invest in tailored blends when safer concentrations will do the job.
Formaldehyde Release and Regulation
Much of the concern around imidazolidinyl urea focuses on its nature as a “formaldehyde-releasing preservative.” Under normal usage, trace amounts of formaldehyde get released—enough to protect against spoilage, but not at levels classed as hazardous by major authorities. Regulatory limits in the EU, US, and Asia exist for good reason. Our regulatory compliance labs track those numbers tightly. We analyze every batch for both purity and release profile. The high-grade raw materials and manufacturing processes we use keep unwanted byproducts beneath permitted levels, and every lot comes with full analytical profiles before it ships to a partner. Water content, pH, and formula interactions all play a part in how much formaldehyde ends up loose in the final use case, and these parameters matter more than raw ingredient percentages alone.
Practical Steps for Safer Formulation
Formulation chemists look at not just label safety, but actual performance. Many companies limit the use of imidazolidinyl urea to lower concentrations, sometimes combining it with milder preservatives to lower the overall chemical load. Lower dosages can still preserve water-based products, which balances safety and effectiveness. Alternative preservatives exist, but switching to unfamiliar chemicals can sometimes introduce other risks, like less robust antimicrobial defense. We regularly evaluate every alternative, knowing that finding a “perfect” solution isn’t as simple as swapping out a single chemical.
Final Thoughts for Those With Sensitive Skin
If you have sensitive skin, scan ingredient lists, check for reputable brands that publish their formulation and safety data, and consult your dermatologist. No preservative is risk-free. Still, modern manufacturing attention to detail, regulatory compliance, and consumer feedback help keep personal care products both clean and widely tolerable. As manufacturers, we continue monitoring new safety research and improving how ingredients like imidazolidinyl urea get used, always with both safety and performance in mind.
Understanding Imidazolidinyl Urea from the Factory Floor
Every day, teams on our manufacturing line track the chemistry behind each ingredient, and that includes the widely used preservative Imidazolidinyl Urea. It’s pretty common in the personal care and cosmetics sector for controlling microbial growth. But there’s more to the discussion: does Imidazolidinyl Urea give off formaldehyde? Here’s a perspective informed by years of hands-on experience handling and analyzing this material in batches ranging from grams to tons.
Real Chemistry and Real Risks
Imidazolidinyl Urea comes from a group called formaldehyde donors. That’s not a label tossed around lightly. The chemistry takes urea and other reactants and bonds them with formaldehyde. In storage or in products, especially under higher pH, heat, or over long periods, it can eventually break down and release some free formaldehyde. In our work, we’ve measured actual levels coming off fresh batches and products aged on the shelf. Trace formaldehyde is no myth—results show that small, but measurable, quantities can be released.
This isn’t a secret in the industry. Regulators know that certain preservatives may introduce formaldehyde. The EU and other regions have set strict limits on how much can appear in finished formulas. It’s not just about following the rules—it influences choices at every step. Even small differences in pH or temperature during mixing can nudge formaldehyde rates up or down. From our perspective, careful control keeps levels below those regulatory caps.
Why Formaldehyde Release Catches Attention
Most questions about Imidazolidinyl Urea come from ingredient transparency. Consumers ask for safer formulations. Concerns center on formaldehyde’s status as an irritant and, at certain exposure levels, a carcinogen. No one in manufacturing wants batches turning out with more formaldehyde than expected. So, the best factories watch for it constantly. In practice, every production lot gets regular tests. Modern analytical technology spots formaldehyde at sub-ppm levels.
The science shows that formaldehyde from Imidazolidinyl Urea is dose-dependent and can fluctuate. If a product designer wants as little as possible, they consider milder preservatives or tweak the formula’s pH and dosage. Some industries push for “formaldehyde-free” claims, but the chemistry behind most donor preservatives means that absolute zero is hard to hit for every formulation.
Solutions from an Insider’s Perspective
In our plant, much of the effort goes into preventing breakdown before the ingredient even leaves the building. Drum storage temperatures stay cool and dry. Blends use as little Imidazolidinyl Urea as technically possible, relying on optimized cleanroom techniques that lower contamination risk so that preservatives can be dialed down.
Regulators may update permissible limits as research evolves. As chemists, we keep an eye on evolving evidence, but also monitor the technical side—new preservative blends, adjustment agents for pH, and alternative technologies. It’s always a balance: shelf life, consumer experience, safety, and staying nimble enough to match new science and rules.
Trace formaldehyde isn’t just a theoretical risk. Any responsible manufacturer recognizes consumer concerns, works to reduce exposure, and runs thorough lot testing, not just to meet the law but to ensure real trust in each batch.
Our Experience Manufacturing Imidazolidinyl Urea
From a manufacturer’s perspective, every raw material tells its own story. Imidazolidinyl urea forms part of this narrative for personal care product makers because it has a long track record as a preservative in cosmetics, lotions, and other topicals. Our team has worked with the substance since the early days of its wide-scale adoption. Over years watching regulatory bodies and customer feedback, one theme stands out: preservatives need a strong safety margin, and the market holds them to a high standard.
Allergic Reactions: How They Occur and What We’ve Seen
Imidazolidinyl urea works by releasing low levels of formaldehyde, which disrupts microbial growth in finished products. The problem is that trace formaldehyde matters, especially for sensitive populations. People with a formaldehyde allergy can react to imidazolidinyl urea at concentrations permitted by cosmetic regulations. Reactions tend to show up as redness, itching, or a burning sensation—typical symptoms of contact dermatitis. These reactions are not the norm, but they show up often enough that professionals recognize the risk, particularly in individuals who already know they react to formaldehyde or “formaldehyde releasers.” From customer support tickets and feedback from our downstream partners, we know that skin reactions remain rare, but the impact for those few individuals can range from inconvenient to unpleasant.
Balancing Safety and Demand in Real-World Production
Testing for safety takes up a big part of our daily business. We follow international guidelines, like those set by the EU and the U.S. FDA, limiting the amount of preservative used. Batch release in our plant always includes a review for compliance on finished product concentration. To lower risks even more, we invest in purity analysis, making sure batches don’t drift out-of-spec on raw material impurities—impurities have sometimes caused problems in the past with unrelated compounds, and the lesson applies here.
Calls for alternatives are getting louder. Dermo-cosmetic brands and private labelers want preservatives that barely register as allergens even for the most sensitive users. New biocide blends and “formaldehyde-free” systems are in development, but they create their own manufacturing headaches: shelf life, cost, physical compatibility with other formula ingredients.
Managing the Human Side: Labeling, Testing, and Credible Information
Honest communication helps the end user to make informed choices. We support full ingredient disclosure and call out formaldehyde risks in technical discussions with our B2B clients. A policy like this means we occasionally steer brands away from imidazolidinyl urea if their end customer base trends toward sensitive skin. Patch testing during formulation goes a long way in reducing incidents, and results from clinical labs guide our commercial recommendations.
Incidents of formaldehyde sensitivity—while uncommon—deserve attention. Professional organizations such as the American Contact Dermatitis Society recommend avoidance for known sensitive individuals, and mainstream regulatory authorities agree.
Looking Ahead: Continuous Improvement and Safer Formulas
The chemical supply chain faces constant adjustment as new research appears. Scientists compile real-life case reports, and manufacturers adapt recipes or look for greener options. Our lab is tracking preservation alternatives that offer the same protection against microbes without contributing to allergy lists. At the factory, we keep documentation transparent, update customers if regulatory positions shift, and invest in analytical methods so clients can verify claims independently.
For anyone developing a skin-contact product today, the biggest win comes from targeted risk assessment, honest marketing, and data-driven decision-making. Through this approach, the industry gets products that work and minimize risk for everyone—consumers, manufacturers, and partners alike.
Real-World Insights From the Manufacturer’s Perspective
Walking into a formulation lab, formulators handle raw Imidazolidinyl Urea nearly every day. The subject of what actually counts as “natural” or “organic” pops up again and again, especially as trends and consumer pressure push toward greener products. The truth is, the chemistry tells a straightforward story—one frequently missed by headlines and clean-label marketing campaigns.
Imidazolidinyl Urea keeps creams and lotions safe from microbial growth. It earns its role by providing reliable preservation in water-based cosmetics, where bacterial contamination would otherwise ruin the product and create safety risks for consumers. The substance appears in countless formulas spanning skin care, personal care, and certain pharmaceuticals. It preserves more than just the formulas—it protects reputations by keeping products from spoiling on the shelves.
Let’s talk about the natural and organic labels seen at trade shows and on finished goods. International standards, including those from major certification bodies like COSMOS or USDA Organic, maintain strict definitions. For organic certification, allowed substances typically appear on a positive list compiled by the certifying body. Imidazolidinyl Urea never finds a place on that list. The manufacturing process relies on synthetic chemistry, starting with petrochemical-derived raw materials. There’s no farm field source, and the molecule doesn’t exist in nature. For those of us running reactors and distillation columns, the synthetic origins of preservatives like this are obvious and unambiguous.
Certifying a formula as natural follows a similar logic. Most certifying groups require that both ingredients and process align with the idea of minimal synthetic alteration. Many certifiers operate a “negative list” of excluded ingredients, and Imidazolidinyl Urea always appears on this list. Even products described as “made with natural ingredients” exclude it if natural certifications are pursued. Occasionally, the debate crops up in the lab: would a lower “dose” or use in a blend pass muster? Certification inspectors and auditors don’t make allowances. Their chemical assessment, grounded in traceability and process, keeps synthetic preservatives out of the running.
This isn’t just a paperwork issue. Years spent in manufacturing taught us the stakes of reliable preservation. We’ve seen small-scale producers turn to essential oils and food-grade alternatives; many struggle to match the broad-spectrum power and stability of Imidazolidinyl Urea. Natural alternatives can work, but their required concentrations risk causing irritation or changing product performance. Process tweaks, more rigorous packaging, and shorter shelf lives offer partial solutions. These work around the absence of robust, synthetic preservatives, but few companies switch without trade-offs in convenience or cost.
Regulators in North America, Europe, and Asia all monitor safe use levels for Imidazolidinyl Urea, setting strict limits in conventional cosmetics. The worldwide consensus excludes it from both organic and natural certification. Anyone promising an “organic” cream with this preservative in the ingredient listing runs afoul of the regulatory facts, not just marketing language.
The call for greener chemistry grows stronger each season. We invest in R&D to explore preservation systems compatible with natural and organic standards, understanding that consumer preferences evolve and regulations will only tighten. For now, Imidazolidinyl Urea’s place is clear—crucial for safety in standard formulas, but off-limits for certified natural or organic claims. Formulators need full transparency about what goes into a jar or bottle and what that means for the product’s true identity in a crowded, competitive marketplace.