Isopropylparaben: An Industry Perspective on a Versatile Preservative
Historical Development
Working with preservatives over decades, we have watched synthetic esters of p-hydroxybenzoic acid, like isopropylparaben, grow from chemistry-lab curiosities into mainstream industrial staples. Parabens entered commercial markets as far back as the 1920s, aiming to fight spoilage when natural options fell short. Early work revealed that placing different alcohol groups on the paraben backbone changed its safety and antimicrobial activity. Isopropylparaben doesn’t ride the coattails of its more famous siblings, like methylparaben and propylparaben. Instead, its rise resulted from the need for balanced preservation: strong enough to control contamination but compatible with sensitive formulations. As hygiene standards rose in food, cosmetics, and pharmaceuticals, producers like us had to step up research, improve synthesis, and resolve toxicity questions. Over many years, we saw the regulatory landscape shift, with authorities scrutinizing safety data and pushing manufacturers to refine both ingredients and processes. The demand for isopropylparaben in applications emerged only as manufacturers and formulators learned to blend efficacy, safety, and regulatory compliance.
Product Overview
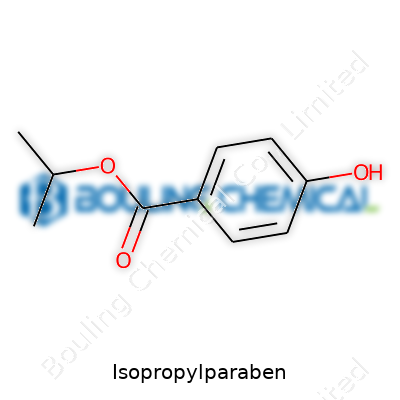
Industrial experience says isopropylparaben looks modest: A white, fine crystalline powder or sometimes a small solid mass, almost odorless, sparingly soluble in cold water but dissolving readily in alcohol and ether. Its identity sits in its structure—4-hydroxybenzoic acid joined with isopropyl alcohol through an ester linkage. The product’s purity and composition drive value, not fancy packaging. In our plants, major concerns often involve batch consistency, absence of trace impurities, and compliance with international pharmacopoeias. If purity strays too far, stability or safety suffers. Operators and quality control teams know that shelf life hinges more on controlling contamination and container selection than on superficial changes in the compound. Customers, whether in cosmetics or pharmaceuticals, expect detailed origin traceability. Open supply-chain practices stem from decades of real-life recalls—nobody in chemical manufacturing willingly gambles with unknowns.
Physical & Chemical Properties
Isopropylparaben melts at roughly 76–78 °C. Unlike some competitors, it doesn’t break down easily at room temperature. This physical durability underlies its value to manufacturers—ingredients that survive transit and mixing limit customer complaints. In practice, its low solubility in cold water demands careful pre-mixing or dispersion to avoid visible residues or analytical issues. Chemists favor the moderate lipophilicity, which supports better preservative action in emulsions and creams than many shorter-chain parabens. Yet, the line between effectiveness and overuse is narrow. The chemical structure grants good shelf stability, so a well-made isopropylparaben batch rarely gives up its preservative qualities for years. Accurate melting-point and assay testing serve as quick barometers for quality, and no manufacturer takes shortcuts with batch-release parameters.
Technical Specifications & Labeling
In manufacturing, technical details are more than box-ticking—they drive everyday processing choices. Isopropylparaben typically contains over 98% assay by HPLC or GC, with strict limits on related substances and residual solvents. Ash and loss-on-drying figures offer clues to undetected contamination. Most markets set strict maximum allowable impurity contents, especially for uses touching food or skin. Workers follow systematic labeling backed by up-to-date safety data sheets, disclosing not only the CAS number and chemical name but also recognized synonyms. It’s not just about regulatory risk—hazard identification impacts how operators store material and react to spills or misuse. Years of audits have proven that hidden corners in technical documentation come back to bite only after an accident or customer complaint, so full transparency serves the whole chain.
Preparation Method
In a production facility, the route from raw material to packaged isopropylparaben captures the industry’s priorities. Most producers rely on esterification: reacting isopropyl alcohol with p-hydroxybenzoic acid, using acid catalysis (often sulfuric acid) under controlled heating. This reaction produces water as a byproduct, which must be continually removed to drive the reaction toward completion. Knowledgeable operators control both the temperature and stoichiometry, monitoring water removal and controlling sulfuric acid levels to avoid over-acidification and unwanted color development. After reaction, neutralization, purification, and careful recrystallization steps protect against residue and odor—all common complaints from finished-goods manufacturers. Purity isn’t just a number; every contaminant left unchecked creates hours (or days) of troubleshooting further downstream. Differences in equipment and process-flow between large and small plants can impact yield and waste, so every adjustment starts from hands-on experience with both chemistry and compliance.
Chemical Reactions & Modifications
As a phenolic ester, isopropylparaben reacts in predictable ways. Hydrolysis occurs in strong acid or base, splitting it into p-hydroxybenzoic acid and isopropyl alcohol—chemistries that mark the basis for stability studies in all new applications. Exposure to strong oxidizers or direct sunlight can degrade the molecule and cut preservation time, lessons learned from off-color or ineffective products in early years. Skillful chemists tweak substitution patterns or introduce functional groups to tailor antimicrobial effects or reduce skin penetration, contributing to safer leave-on formulations. Such modifications often respond directly to evolving regulatory or market pressure, with chemical manufacturers supporting R&D teams in real-world pilot batches. Proprietary derivatives and blends often originate from problems solved on the fly during past projects, not from textbook recipes.
Synonyms & Product Names
Industry documents refer to isopropylparaben using several names: 4-hydroxybenzoic acid isopropyl ester, or even para-hydroxybenzoic acid isopropyl ester, sometimes abbreviated as IPPB or E216 in certain contexts. Each reference signals a need to navigate both global and local regulatory codes. Misnaming or imprecise labeling causes major confusion both in supply chains and in regulatory filings, so manufacturers maintain updated synonym lists and screen for outdated terminology. A batch held up by customs or import authorities over paper errors costs more than any added labeling step. Coordinating with international product registries and standards committees has become a daily fact of life in global manufacturing.
Safety & Operational Standards
Years of plant operation reinforce simple truths: safe handling practices and detailed hazard information dictate both insurance costs and the well-being of production crews. Isopropylparaben’s risk is moderate—dust inhalation or skin contact irritates, but flammability risk stays low under normal conditions. Regular training, local exhaust ventilation, and personal protective equipment remain routine because onset symptoms sneak up quickly. Safe storage demands sealed containers and dry, cool areas, even if the bulk material displays admirable physical stability. Companies perform incident drills for spills and accidental exposures, and every batch must ship with certificates of analysis and traceable safety data sheets. National and international chemical safety standards impose documentation and strict limit tests, driving investments into environmental controls, automated dispensing, and electronic batch records.
Application Area
Across cosmetics, pharmaceuticals, and certain food contexts, isopropylparaben acts as a shield against bacteria and molds. Smaller producers have noticed shifts in consumer preferences—calls for lower inclusion levels, avoidance of certain parabens, or “clean label” claims. Large manufacturers must react by either pursuing alternative preservation systems or by supplying grades of isopropylparaben with higher purity and validated safety documentation. In pharmaceutics, stringent good manufacturing practice regulations demand not only documentation of content but also of potential leachables and extractables. In oral-care and personal-care products, companies adapt formulations based on regional regulatory approvals or restrictions. No less important, pilot-scale trials often uncover subtleties in ingredient compatibility that generic monographs or literature can miss. These hard-won insights get passed between R&D and manufacturing until workable solutions reach commercial scale.
Research & Development
Research teams targeting next-generation preservation constantly explore structure–activity relationships, the interactions between mushrooming regulatory guidance, and new analytical detection limits. Our own collaborations with universities and clients have shown that adjusting alkyl groups or pairing isopropylparaben with chelating agents can stretch shelf lives or reduce total preservative content. Analytical chemists track residue levels down to parts per billion, using advances in chromatography and mass-spectrometry. Development can’t remain static, given not only regulatory churn but also evolving pathogens, skin microbiome science, and consumer skepticism. We invest in cross-disciplinary R&D, integrating feedback from end-users and drawing on data from post-market surveillance. Overlooked research—such as how packaging materials interact with paraben esters—regularly generates real commercial value.
Toxicity Research
Toxicological evaluation shapes ingredient status more forcefully than any marketing campaign. Findings on isopropylparaben establish relatively low acute toxicity, and it seldom triggers severe irritation at the concentrations typically used in cosmetics and pharmaceuticals. Research on reproductive and endocrine-disrupting effects has stirred the global debate, leading to periodic reassessment of permitted concentrations. Chronic exposure studies often show wide margins of safety, but manufacturers prioritize ongoing surveillance, submitting fresh toxicological data and reviewing real-world incident reports. Long-term animal studies, cell-culture assays, and human patch-tests together construct the safety database. Regulatory authorities worldwide monitor data closely, and any newly identified risks prompt swift review of usage levels and labeling. Collaboration with independent toxicologists gives companies the data credibility needed for global registrations and market access.
Future Prospects
Looking ahead, manufacturers see both challenge and opportunity. Regulatory trends continue to tighten, forcing a rethink of both product design and quality-assurance systems. Demands for “green chemistry” solutions and biodegradable alternatives directly compete with the efficiency and broad-spectrum activity of traditional parabens. We anticipate stronger restrictions on maximum permitted levels in some regions and increased documentation requirements almost everywhere. At the same time, antimicrobial performance in personal-care and medical applications will stay critical. Companies benefit by investing in cleaner production technology, exploring new delivery systems for preservatives, and supporting transparency throughout the supply chain. Collaborations that connect industry, academia, and regulatory bodies remain key to balancing public health, consumer demands, and real-world manufacturing practicalities. Our experience tells us that adaptation—not static reliance on old recipes—secures both regulatory compliance and market trust.
Understanding Isopropylparaben’s Role
Isopropylparaben falls into the family of parabens, known for their preservative properties. We see it most often in personal care, cosmetics, and some industrial applications. Our batches head out to customers looking to protect their formulations from spoilage—bacterial or fungal growth can ruin both consumer trust and economics. Isopropylparaben acts as a reliable shield against such problems. Working directly in manufacturing, we see quality control teams depend on its wide pH stability. Even at low concentrations, it limits microbial growth, which helps avoid recalls, product instability, and waste.
What Drives Its Use in Products?
Cosmetics factories want their creams, lotions, and deodorants to last—nobody likes a product that spoils in the cabinet. Microorganisms thrive in water-based formulas, especially once packaging is opened. Our customers need preservatives that won’t shift a product’s texture or odor, and Isopropylparaben fits that need. It works well with other parabens, giving manufacturers flexibility in how they design their antimicrobial defense. This ability to blend with partners like methylparaben or propylparaben means formulators can tackle a broader range of microbes without risking skin irritation at high single-preservative doses.
Pharmaceutical clients choose Isopropylparaben for topical products, ointments, and certain liquids. The logic runs parallel—microbes can’t thrive, so shelf life lengthens. Cutting spoilage lowers overall cost and waste, which is as important as upfront formulation decisions. We watch finished product lots undergo regular stability testing, and Isopropylparaben stands up under stress so the product doesn’t break down before a customer uses it all up.
Safety and Perception
Safety standards run high on substances that touch skin. Regulatory agencies across the globe monitor paraben use and require manufacturers to keep use levels below internationally accepted thresholds. The scientific literature signals that, within regulated amounts, Isopropylparaben works safely. As a manufacturer, we track research, examine batch traceability, and provide full documentation packages to downstream clients. Open data builds trust in our chemical’s consistent performance. Over the last decade, consumer awareness about ingredient lists drove companies to weigh alternatives and increase transparency. This hasn’t knocked out Isopropylparaben but shaped how brands communicate about it.
Addressing Concerns and Future Directions
Voices have aired health and environmental concerns about parabens. The reality on the factory floor looks more nuanced. We support responsible use, never encouraging levels above regulatory guidelines. On the environmental side, reformulation toward preservation systems with renewable feedstocks or improved biodegradability is ongoing. Chemistry advances don’t happen overnight, but steady R&D work is part of our routine—pilot trials, alternative molecules, and customer input all feed continual improvement. As more data emerges, we stay responsive, keeping an eye on ways to meet safety and sustainability expectations without sacrificing protection against microbes.
Final Thoughts from the Production Side
Experience in chemical production shows that Isopropylparaben remains a workhorse in product stability. It guards value along the supply chain from the very mixing tank through transport to the consumer’s shelf. Our commitment holds steady: tight quality controls, data-driven safety, and honest dialogue with customers about ingredient choices. As expectations evolve, so does our approach—balancing proven chemistry with progress toward greener preservative solutions.
Our Perspective as a Chemical Manufacturer
The debate around isopropylparaben in cosmetics keeps resurfacing. As a chemical manufacturer with years of industry experience, we’ve watched this ingredient move in and out of favor. Isopropylparaben acts as a preservative, discouraging the growth of bacteria and mold in creams, lotions, and shampoos. Without preservatives, these products spoil quickly and turn into a breeding ground for microbes. This reality highlights why chemists consider certain parabens for cosmetic use, despite ongoing scrutiny.
Regulatory bodies around the world do not view all parabens the same way. Some, including isopropylparaben, have faced restrictions or outright bans in regions like the European Union due to studies looking at potential hormone-disrupting effects. Animal testing on high doses sparked those concerns, but those tests do not reflect the trace amounts found in finished consumer products. Chemicals used at trace levels in a finished formula behave much differently from larger, isolated doses. Our formulation chemists test every batch to verify the actual levels fall well below accepted safety margins.
In the United States, the Food and Drug Administration recognizes parabens, including isopropylparaben, as safe for use within established limits. Their position results from decades of product monitoring and toxicological assessments. The Cosmetic Ingredient Review, an independent panel, came to a similar conclusion. They evaluated exposure, skin absorption, metabolism, and detected no compelling link between parabens and health problems at levels used in cosmetics.
Even so, public awareness keeps shifting. Retailers and brands respond by marketing “paraben-free” products, raising questions among everyday users. As chemists, we know removing isopropylparaben raises new preservation challenges. Without robust alternatives, companies often add other synthetic ingredients that still present safety and allergy risks. These swaps do not always deliver better outcomes for users or the environment.
Our research and quality control teams run stability studies on product lines, both with and without isopropylparaben. We often find that removing this preservative shortens a product’s life on store shelves. Spoilage risk rises, and recalls become more common. The result: more waste and higher prices for consumers. In some markets, brands then import formulas from countries where isopropylparaben remains on the approved list. This global patchwork leads to confusion for buyers and formulators.
The pursuit of safer beauty products never ends. We believe safety lies not in demonizing one ingredient or favoring another, but in making decisions based on real exposure levels and reliable science. Toxicology always depends on dose, route of exposure, and how ingredients interact within the final formulation. Modern manufacturing methods and advanced analytical tools help us keep contaminants and impurities far below regulatory limits.
Our teams stay alert to new findings. We invest in transparent labeling and clear technical documents so brands and consumers know exactly what’s in each product. If local laws or scientific consensus change, our production lines adapt.
Making cosmetics both safe and effective means understanding each ingredient’s strengths and trade-offs. Isopropylparaben serves a role in keeping products fresh and free from harmful microbes. The conversation about its safety should focus on facts, measured risk, and open communication—not just trends or marketing claims.
The Reality of Isopropylparaben from a Manufacturer’s View
Working hands-on in the chemical production of preservatives, we have seen every question tossed our way about safety and tolerability. One of the most discussed ingredients, Isopropylparaben, comes under the microscope every year. Many consumers worry about potential for allergies or irritation in personal care and cosmetic products. Through actually making and testing batches, not just moving barrels, we spot patterns and see what really comes up during use and regulatory review.
Understanding the Ingredient and Its Track Record
Isopropylparaben is part of the paraben family, which got its main use for protecting lotions, shampoos, and creams from harmful microbial growth. Over decades, regulatory agencies have sifted through data on parabens, checking everything from sensitization to outright allergy. Even now, we test for purity, residue, and potential contaminants in every lot we produce, following established methods like those set out by the European Pharmacopeia. Consistency matters. No company can hide behind a spec sheet if their customers report burning or itching — those messages reach us quickly.
Most reactions studied have not centered on Isopropylparaben itself. Methylparaben and Propylparaben, the more widely used types, draw more research and case reports of allergic contact dermatitis. Isopropylparaben lands in the lower usage group, so direct clinical evidence appears less often. From the manufacturing floor, requests for this ingredient now usually emphasize purity and trace removal of possible byproducts, just in case someone reacts to substances besides the main compound.
Biology Behind Reaction Risks
Everyone’s skin tells a different story. Some react to almost anything, even aloe or vitamin creams. Actual allergic response to parabens stays rare in published findings, documented at rates well below one percent among patch-tested patients. Typical skin reactions, especially redness or itch after using a preserved product, often trace back to fragrances or breakdown from old stock on the store shelf. We routinely keep our batches under tight control for shelf-life and storage conditions for this reason.Strict tracking lowers the risk of breakdown products that do irritate sensitive users.
How We Address Concerns Directly
Feedback from downstream clients shapes our process. Larger personal care brands ask for certificates of analysis with every drum. We support them with ongoing transparency about production process changes and quality assurance audits. On rare occasions—if a buyer reports complaints about redness after product launch—we always ask for the raw batch code. Cross-referencing this code with our own logs allows us to confirm raw material status at the time of manufacturing. After reviewing final preservative concentration and test results for purity, we often discover that the real source of irritation lies elsewhere in the formulation, or in outside environmental factors.
Looking Toward Safe Use
Parabens, including Isopropylparaben, continue to go through global safety reviews. Multiple bodies like the Scientific Committee on Consumer Safety and U.S. FDA review the latest data. Many countries have set legal limits on maximum allowable content of parabens for a reason — years of toxicological studies have shown a wide margin of safety at low doses. Manufacturers like us use these limits as a cap, not as a goal, so most products hit the market far below the upper regulatory threshold. Nothing sits beyond question, and allergy is always possible for any ingredient, natural or synthetic. From experience, keeping communication clear down the supply and manufacturing chain avoids the bulk of problems—before product ever lands in a consumer’s hands.
Understanding Daily Exposure
At our plant, we understand how important it is to be clear about the chemicals we produce, especially those used in cosmetics and pharmaceuticals. Isopropylparaben catches many questions because it functions as a preservative, preventing the growth of mold and bacteria in everyday products like lotions, shampoos, and creams. Parabens have a long track record of use, but public scrutiny has increased over the years.
What Science Has Told Us
Our chemists have reviewed both in-house test data and published science. Health concerns around parabens, including isopropylparaben, center on their potential to disrupt hormones, particularly by mimicking estrogen. Some lab research shows that certain parabens can weakly bind with estrogen receptors in isolated cell studies. That’s the science that led to regulatory attention, especially in Europe, where authorities placed concentration limits on several parabens used in leave-on products designed for children under three.
These legal limits come from a precautionary approach. In practical terms, most studies found that isopropylparaben gets broken down quickly and doesn’t linger in the body. Typical exposure from skincare falls well below thresholds that toxicologists associate with possible effects in animals. In our experience producing this compound, trace residues dissipate fast, even before consumer use.
Regulation and What It Means for Users
Our regulatory team keeps up with both North American and international updates. At the moment, the United States Food and Drug Administration allows isopropylparaben in cosmetics without specific restrictions, though it keeps ongoing surveillance on new published research. The European Union restricts use of isopropylparaben in personal care items to a maximum concentration to cap total exposure, part of a broader move to limit cumulative parabens in sensitive populations.
Several large brand formulators request assurance and full traceability from raw materials through finished ingredient. We often support audits and documentation to verify clean routes of synthesis, tight batch controls, and absence of unlisted contaminants. Our teams answer questions on whether this preservative forms impurities such as p-hydroxybenzoic acid, and production always follows the strictest standards on recordkeeping and environmental protection.
Looking Forward: Listening and Design Choices
As a chemical manufacturer, we know the discussion about preservatives is far from settled. Some brands have moved away from parabens, often based on consumer demand rather than strict science. Our approach is to inform formulators of the latest safety data, offer high purity materials, and be transparent about what’s known and what’s still being debated. Whenever a new toxicological alert or regulatory shift appears, our people quickly investigate possible substitutions and communicate the real-world impact this change would cause on performance, shelf life, and price.
Looking beyond compliance, the best defense against health risk is strong quality control at every stage. Regular improvements in synthesis and stricter testing standards have cut the chance of unexpected side products. As new data comes out, we plan new studies and stay in close touch with both independent labs and trade associations. Honest discussion, scientific accuracy, and public trust shape our day-to-day operations as much as any technical process does.
Regulations Drawn by Real-World Safety Concerns
We manufacture isopropylparaben for use in numerous cosmetics and personal care products. In recent years, our industry has faced plenty of questions about its legal status in various countries. Most of these regulatory shifts come from safety reviews, consumer pressure, and evolving global guidelines. Our technical teams pay close attention to these developments since they determine where our isopropylparaben products can be supplied.
Where the Ban Actually Exists
European regulators reviewed isopropylparaben along with several other parabens. In 2014, the European Commission officially banned it for use in cosmetics under Regulation (EC) No. 1223/2009. This wasn’t a surprise—long-term safety data on isopropylparaben was lacking, and regulators in Europe often set the pace for ingredient scrutiny. The main worry has been its possible hormone-disrupting properties, though concrete evidence is limited.
So, in Europe, you cannot put isopropylparaben in cosmetics or toiletries at any concentration, not even for restricted use. Regulatory authorities followed their precautionary principle: when conclusive proof of safety is absent, the door closes.
Other markets, such as the United States and Japan, have taken a different path. Agencies in those regions have not imposed any outright bans. They maintain close observation of emerging safety studies, leaving the decision to individual manufacturers and brands, so long as the ingredient amount follows sensible guidelines and does not cause skin reactions in normal use.
Industry Impact: Downstream Adjustments
We have felt the weight of the European ban. Before 2014, European customers made up a significant share of our isopropylparaben sales. That changed overnight once the legislation became enforceable. Our production lines in Asia and North America had to adjust packaging, change documentation, and even reformulate blends to eliminate isopropylparaben for exports aimed at the EU. This ban also triggered certain multinationals to move away from parabens altogether, even in places where they remain legal, to avoid relabeling and litigation headaches.
Developing substitute preservatives has proven complicated. Few alternatives deliver the same antimicrobial protection without side effects or unstable shelf-life. When switching out isopropylparaben, manufacturers tend to run more stability testing and micro-challenge studies—which adds time, cost, and sometimes brings disappointing results.
Looking Forward: Navigating Safe and Legal Preservation
Highly regulated markets like Europe can push new ingredient development, but they also remove a versatile option from formulators' toolkits, making compliance costlier at every step. As a manufacturer, continuous research sits at the center of our mission. We partner with labs and ingredient suppliers to update safety dossiers and document data as it emerges. Transparency and responsiveness have started to matter more than ever: customers, whether end-users or OEMs, ask tough questions about every preservative material.
Science can change old assumptions, but national bans stay in place unless regulators see robust, peer-reviewed safety data to reverse them. As it stands, isopropylparaben remains legal in most non-European markets, but any company involved in global supply chains should verify each country’s latest position before using it. Until clearer international alignment develops, the industry keeps balancing regulatory trends, consumer sentiment, and the realities of safe formulation.