Insights on Methyl o-benzoylbenzoate: Perspective from the Production Floor
Historical Development
Methyl o-benzoylbenzoate has roots stretching back to the early 20th century, landing on the radar of synthetic chemists who searched for new photoinitiators and intermediates for pharmaceutical and material science applications. It entered regular industrial synthesis as technical methods advanced, especially through advances in Friedel-Crafts acylation and esterification techniques. Over decades, researchers refined catalyst systems and reaction parameters, trimming away inefficient byproducts and raising yields. Watching the evolution from gram-scale university bench work to large-scale reactors in purpose-built chemical manufacturing facilities, we learned a lot about managing heat transfer, acidity, and downstream purification. Knowledge built up through experience, not just literature. We can trace the nods to Methyl o-benzoylbenzoate in published patents and company research reports; those drew boundaries for scaling quantities safely and efficiently and for achieving reliable batch-to-batch analytical results. Technical advancements pushed cost-per-kilogram down and supported more diverse product grades, responding to new industry demands rather than relying on a one-size-fits-all approach.
Product Overview
Methyl o-benzoylbenzoate typically emerges from our reactors as a nearly colorless to pale yellow crystalline solid. Staff orange and yellow flakes apart all too often, a sign that temperature drifted during crystallization or that solvents from earlier steps contaminated the process. By sticking close to analytical testing and hands-on observation, our factory crews learned what operations produce the cleanest output. Its value in industry comes from both its reactivity and consistency. Many clients in the coatings, adhesives, and UV-initiated systems rely on reproducible quality, which means our Q.C. teams look for trace-level impurities. The compound’s not an end in itself for many clients; instead, it acts as a stepping stone for further modifications or a backbone in more complex formulations.
Physical & Chemical Properties
Methyl o-benzoylbenzoate brings together essential features—a crystalline solid in standard conditions, with a melting point accessible to basic industry steam or hot water systems, and moderate solubility in solvents like acetone, dichloromethane, and toluene. Its faint aromatic scent shows a reassuringly low volatility, which reduces risks of worker overexposure and limits losses during storage and handling. Incompatible conditions or contamination show up quickly as altered color, loss of purity, or unexpected exotherms in the reactor. Hands-on experience showed us that this compound resists hydrolysis better than many similar esters, as long as it's kept away from strong bases or acids in storage. The most critical property, from the perspective of users in UV-curing resins, is the absorption spectrum: the aromatic rings pull in ultraviolet light in a well-studied range, setting it apart as a favored photoinitiator or modifier. Our long-run product consistency means equipment doesn’t need constant recalibration for trusted partners.
Technical Specifications & Labeling
Product specs for Methyl o-benzoylbenzoate in our plant emphasize real-world outcomes, not theoretical maxima. We focus on minimum assay requirements, color standards, trace impurity profiles, and water content, all confirmed with well-calibrated chromatographic and spectroscopic analysis. Labels on drums, bags, and other containers make it clear: batch number, net weight, production date, and hazard category. Handling instructions align with our on-site experience rather than just regulatory summaries—we see first-hand what happens when overlooked moisture content or mistaken labeling causes spoilage, caking, or regulatory headaches. Lessons learned from near-misses pushed us to design labeling layouts that work under bright lights, in humid conditions, or by workers wearing protective gear, rather than assuming optimal conditions. Our technical certifications track not only regulatory code compliance but also specific client needs established over long relationships. Documentation proves its worth when containers move globally and customs officials demand credible, transparent data.
Preparation Method
The route we use for Methyl o-benzoylbenzoate starts with ortho-benzoylbenzoic acid, sourced from aromatic hydrocarbon feedstocks refined through Friedel-Crafts acylation. After extracting and purifying the acid, we react it with methanol in a controlled esterification, using acid catalysts chosen for high conversion and manageable side reactions. Corrosive environments demand careful material selection—reactor linings, heat exchangers—because mistakes lead to downtime and unnecessary costs. Our decades of batch-processing experience mean we can adjust charge ratios, catalyst loadings, and process temperatures based on product performance data, not just standard operating procedures. Careful washing, recrystallization, and solvent stripping keep product color and purity in tight windows, since customers picking up visible shifts will flag more than just QA paperwork. Decades of pilot batch and full-scale operations taught us to expect unexpected bottlenecks—from feedstock inconsistencies to scheduled utility outages—prompting process adjustments without risking quality or safety.
Chemical Reactions & Modifications
Our years of hands-on work showed where Methyl o-benzoylbenzoate goes in industrial labs. It stands up to further esterifications, reductions, and acylations, offering functionalization sites at the aromatic rings and the ester group. Many downstream users count on clean, high-yield modifications—hydrolysis to the parent acid under controlled alkaline conditions, amidation to offer biologically active structures, or halogenation to create new photoinitiator families. Each pathway results from careful optimization, not just textbook chemistry. The experience of managing real-world batch reactions at scale leads to better planning around heat evolution, byproduct management, and product isolation. In the fast-moving world of specialty chemicals, R&D efforts often request gram or kilogram batches of functionalized derivatives. Feedback from these users guides our approach, leading us to adjust reaction windows, purification steps, or packaging forms to address unexpected challenges.
Synonyms & Product Names
Names matter on the production floor and throughout supply chains. Methyl o-benzoylbenzoate crops up as MBB, 2-benzoylbenzoic acid methyl ester, and methyl 2-benzoylbenzoate. Old technical literature, user requests, or regulatory paperwork sometimes create confusion, so clear communication and provision of all synonyms help us avoid missed connections or cross-shipments. As global trade picks up pace, regional regulations and language differences increase the need for transparent cross-references, and mistakes in naming can trigger customs delays or end-user quality complaints. Our logistics crew finds that plain language labeling paired with established CAS numbers prevents the majority of headaches, but knowledge of older trade names avoids costly misidentification.
Safety & Operational Standards
Years of handling, shipping, and processing have deepened our understanding beyond broad regulatory bulletins. Methyl o-benzoylbenzoate does not ignite easily, but dust formation means explosion-proof safeguards, static controls, and high-pressure venting shouldn’t be skipped. Though oral and dermal toxicity remains in the moderate range, inhalation and dust contact can irritate skin and eyes, especially after long exposure on hot, humid days where PPE compliance slips. Our operations team built a safety culture that treats near-incidents as learning opportunities—quick spill cleanup, regular ambient air checks, and real-world emergency drills taught by experienced shift leaders. Waste handling doesn’t stay an afterthought; inappropriate mixing with strong bases or burning without scrubbers leads to corrosive or toxic emissions and requires diligent disposal standards. Standards evolve, so ongoing training covers fresh findings, not just static rules.
Application Area
Most of what we ship flows into UV-curing inks, coatings, and adhesives. The electronics sector uses it for specific photoresistant polymers where edge contrast and crosslinking efficiency must remain sharp. A slice of production goes toward pharmaceutical synthesis, including research into non-steroidal anti-inflammatory drugs and other bioactive compounds. Client collaboration leads us to develop new blends or grades when processing equipment evolves, or when regulations squeeze out legacy alternatives. Our relationships with customers run deep: they share feedback from their own production lines that shapes our process adjustments. From flexible packaging films to niche specialty coatings, every new application points to an industry moving away from commodity chemicals and demanding products that perform to ever-tighter tolerances without hidden variability.
Research & Development
Our R&D division doesn’t work in a bubble—feedback from plant operators and client engineers guides everything we attempt. We keep pushing toward routes that save energy, limit waste, and offer higher selectivity, especially with rising energy and raw material costs. Process intensification, smarter inline monitoring, and introductions of greener catalysts all grew out of honest review of plant bottlenecks and raw material pricing disruptions. Innovations are slow to roll out in batch production, given legacy infrastructure and a culture of safety-first, but persistence pays off. Sometimes, better solvent recovery or a tweak in crystallization temperature brings down batch times enough to free up capacity, opening room for small-lot R&D lines. Academic contacts and participation in cross-industry consortia give us early access to modification ideas, alternate feedstocks, or applications that might seem unconventional now but often point to tomorrow’s standard practice.
Toxicity Research
The body of toxicity data on Methyl o-benzoylbenzoate grew slowly at first. Early animal studies raised some red flags around acute and chronic exposure. Over the years, data from standardized OECD test batteries started to clear up old uncertainties. We follow any new publications closely because risk perceptions can change regulations with little warning. In practice, worker health takes priority—area exposure monitors, updated PPE protocols, and routine health checks became the norm in our shop. Wastewater and air emission data are tracked in real time, not just sent to regulators once a year, and any deviation gets an immediate response. We invest in periodic third-party audits and keep samples archived, so long-term biological monitoring gets support from hard data. Long gone are the days of ‘acceptable’ uncertainty; our teams share safety best practices both inside and between sites to keep trust and transparency at the center.
Future Prospects
Shifts in customer needs push us to keep moving. Demand for photoinitiators in 3D printing, specialty adhesives, and electronic device fabrication keeps climbing. Sustainability pressures—regulatory, customer-driven, and from our own workforce—demand routes that cut hazardous waste and energy use. Upstream, more feedstock variability and the potential for renewable aromatic building blocks shape our research priorities. We see increasing calls for specialty grades—ultra-low impurity, custom particle size, or tailored physical properties—reflecting a world moving toward more complex chemistries and precise performance targets. Facility expansions and capital investment now focus on modular upgrades and process digitization, not more of the same. We keep a close watch on toxicity and regulation reports, ready to pivot or even sunset outdated products in favor of safer, cleaner alternatives that can carry our reputation forward.
Unpacking the Uses from a Manufacturer’s View
Over the years in chemical manufacturing, one molecule that often prompts questions is methyl o-benzoylbenzoate. Inside our plant, batches of this compound roll off the line not just for laboratory orders but for large-scale industrial customers with highly specific needs. The key to understanding the value of methyl o-benzoylbenzoate starts with where it goes after it leaves our facility.
Building Blocks for Photoinitiators
Most requests for this compound come from manufacturers producing photoinitiators. These are specialty chemicals responsible for starting polymerization under UV light, widely used in inks, coatings, and adhesives. We see this in practical terms: factories producing UV-cured products rely on a consistent supply. Methyl o-benzoylbenzoate allows these plants to meet high-speed production targets without sacrificing product quality. The chemistry here isn’t for show—instead, it delivers real improvements in drying times, which directly impacts manufacturing capacity and cost control.
Practicality in Synthesis
Research centers and R&D teams also depend on this compound as a synthetic intermediate. Unlike highly volatile substances, methyl o-benzoylbenzoate behaves predictably during handling and reactions. Researchers trust its stability when testing new routes for more advanced chemicals, including pharmaceuticals and specialty polymers. In our own labs, we have fine-tuned production so that researchers don’t need to worry about batch variability or impurity profiles derailing experiments. Our experience with purification methods lets us keep the active content high and residual solvents low.
Meeting Quality Standards — Not Just Regulations
Regulatory compliance sets the floor for chemical manufacturing. We expect more from ourselves and watch the broader market to keep our processes in line with customer expectations. For photoinitiator production, even trace impurities can lead to residual color in inks or slow reaction rates in adhesives. Years of feedback from real-world customers have shaped our approach: ongoing investment in analytical testing, continuous updates to process control, and strict traceability from raw materials to finished goods. The trust built through consistency matters as much as the paperwork we file for compliance checks.
Sustainability Considerations
Few chemicals are immune from scrutiny regarding environmental impact. The solvents used in methyl o-benzoylbenzoate synthesis and purification require careful management. We recycle where possible and invest in solvent recovery systems. Our plant management monitors emissions and waste output, working with local authorities to stay within safe margins and seek improvements. Customers in the coatings industry often ask about this aspect, looking for suppliers who share their long-term view on sustainability. We believe in supporting not just today’s businesses but tomorrow’s environment as well.
Addressing Current Challenges
Supply chain disruptions have underscored the importance of localized sourcing and reliable logistics. Over the last few years, our team worked closely with upstream suppliers to secure main precursors for methyl o-benzoylbenzoate, reducing unnecessary risks tied to global shipping delays. Open communication and transparent scheduling keep our customers informed, so they can plan production lines without unpleasant surprises.
Looking Forward
The demand for specialty chemicals like methyl o-benzoylbenzoate will keep evolving, especially as digital printing, advanced composites, and next-generation adhesives set new performance standards. For a manufacturer rooted in experience, adaptability paired with quality gives customers confidence. Our role isn’t just to deliver a drum filled with product—it’s to anticipate industry needs, support innovation, and uphold standards that move the business and technology forward.
Understanding the Core Structure
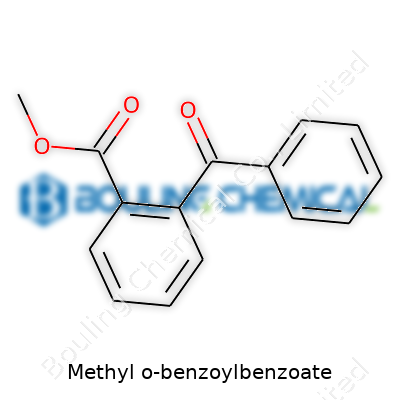
People often ask what makes up methyl o-benzoylbenzoate. This chemical features a methyl ester group attached to an o-benzoylbenzoic acid backbone. To break this down, picture a benzoic acid ring with another benzoyl ring secured at the ortho position. Where you would expect a carboxylic acid, that’s replaced with a methyl ester. That simple swap transforms the molecule for various uses in our industry.
We start with o-benzoylbenzoic acid, where one benzene ring carries a benzoyl group at the ortho position. Methanol replaces the hydrogen from the acid function, so the formula turns to C15H12O3. That gives a crystalline solid with a solid aromatic character. Aromaticity sets up strong chemical stability, which matters for downstream syntheses.
Why Structure Matters to the Manufacturer
Benzoylbenzoate chemistry isn’t just textbook knowledge; it affects every day on the plant floor. In our experience, the structure allows for straightforward synthesis routes; most use a Friedel-Crafts acylation to attach the benzoyl group, then convert the acid to the methyl ester. Every step faces scrutiny because unwanted by-products can complicate purification and set off compliance alarms, so the chemistry shapes our workflows.
The ortho position makes the molecule less reactive than a para version, changing which catalysts and solvents give the best yields. Many teams look at ortho-selective catalysts and fine-tune temperatures to avoid unwanted isomers. Too much acidity or an off-ratio of reactants stalls the reaction — we watch those numbers closely to avoid solvent waste and extra distillation cycles.
Applications Driven by Structure
Aromatic esters like methyl o-benzoylbenzoate work their way into UV-curing systems, colored resins, and specialty intermediates for pharmaceuticals. The rigidity and stability from the double aromatic rings help in polymer cross-linking. The methyl ester opens up options for further reactions, such as transesterification or amidation, used by formulators designing new photo-initiators or stabilizers.
From the manufacturing line, handling methyl o-benzoylbenzoate involves paying close attention to its melting point and solubility. Its chemical structure sets those properties. We use these details to optimize solvent systems for cleaning reactors or cleaning up product after synthesis. Its relatively low toxicity compared to alternatives matters, since operators notice fewer issues with overexposure during handling.
Challenges and Solutions
As demand grows for clean downstream reactions, the purity of methyl o-benzoylbenzoate matters more than ever. Even slight impurities, often by-products from incomplete esterification, cause headaches for polymer and fine chemical producers. We address this using continuous-flow reactors to smooth out temperature swings and keep ratios in check. Column chromatography remains a reliable solution, though we invest in efficient crystallization techniques to minimize solvent use and lower environmental impact.
From the plant’s perspective, a molecule like methyl o-benzoylbenzoate isn’t just a label on a drum. Its structure guides how it’s made, purified, and shipped. Experience shows each aspect — from the dual ring system to that single methyl group — changes everything from efficiency to safety, keeping operations running smoothly and quality consistently high.
Eyes Wide Open: Lessons from the Manufacturing Floor
Every day on the line, the team handles chemicals like Methyl o-benzoylbenzoate. This isn’t one of those compounds you can handle with a ‘business as usual’ mindset. The compound serves as a specialty raw material, often in fine chemical synthesis. Workshop experience has shown the vapors carry a definite risk of eye irritation. Staff who skip goggles, even for a minute, run the risk of stinging, watery eyes—nobody forgets the lesson after that. Basic protective eyewear shields from accidental splashes as well as lingering vapor. Not once have we seen seasoned operators ignore eye protection during transfer or sampling. Forget about trying to ‘tough it out’: direct contact burns and irritates worse than most expect.
Skin Is Not Armor
Lab coats and gloves, not street clothes and open skin, stand as essentials every shift. We’ve tested many different disposable gloves—nitrile holds up the best. Even with this, double-gloving during charging or draining processes has made a difference more than once. Contaminated skin can carry a lingering odor and the compound’s slow-to-fade sticky feel, making washing up a much bigger issue at the end of the day. A recent shortfall in glove supply reminded us: substitute materials do not cut it—proper gloves do. Shortcuts create longer headaches.
Respect for Breathing Zones
Solid ventilation decisions at the plant work better than any mask after exposure happens. Our engineering team assessed and installed local exhaust where open transfer takes place. We rely less on general room air flow and more on targeted capture systems—this greatly reduces the number of complaints about headaches and scratchy throats. Respiratory PPE stays on hand for maintenance work in confined areas. It’s not a luxury. The few times someone thought a quick fix wouldn’t need these precautions, the after-effects proved otherwise. Solvent-like odors mean you’ll know right away if you have a problem, but by that point, mitigation is harder. Real-life experience backs up what the safety data sheets spell out: controlling exposure from the outset protects the whole crew.
Chemistry and Fire Hazards
Storage and handling go wrong when containers are left open or stored near heat sources. The compound itself, though less volatile than some organics, reacts poorly in direct sunlight or close to ignition points. Forklifts have caused more close calls than chemistry itself, so our team minimizes traffic through storage aisles after repeated near-misses. We use grounding straps routinely and we train our newer staff to check every drum for damage or leaks. Near the packaging hall, every spill triggers a protocol—absorbent material, then a careful clean-up, always followed by a check for residue. It only takes one spark, so we keep clean zones rigorously enforced.
The Culture Carried by Experience
We don't assign chemical safety as a checklist—our approach comes from years watching things go right and wrong. Crew members communicate hazards at every shift change, never assuming someone knows the current status. Old-timers know that PPE and ventilation buy security, not just compliance. New staff quickly learn no one laughs off shortcuts. Field experience sorts out recommendations that actually work from those that only look good on paper.
Mistakes with Methyl o-benzoylbenzoate rarely remain small: they turn into training stories passed down for years. In the world of real manufacturing, respect for the risks isn’t theoretical. Everything from the choice of gloves to how drums are stacked reflects lessons written in spilled product, stinging eyes, and, occasionally, a trip to the on-site nurse. We build improvements from these moments, adjusting handling protocols so that new hires never have to learn lessons the hard way. Safety comes from the ground up, not from policy binders.
A Manufacturer’s Look at Responsible Storage
Years in chemical manufacturing drive home some basic truths—chemicals only perform as well as the conditions under which they’re stored. Methyl o-benzoylbenzoate stands as no exception. Like many aromatic esters, this material brings both promise and risks that trace back to physical protection. Skipping even a simple precaution can bring headaches in quality or safety. Our experience synthesizing, refining, and handling bulk batches gives us a clear view of what proper storage means—not abstract guidelines, but tangible impacts on worker safety and customer satisfaction.
Why Storage Location and Temperature Matter
Methyl o-benzoylbenzoate looks stable at first glance and, truthfully, it holds up under sensible conditions. Direct sunlight on drums changes that equation. Ultraviolet light heats surfaces and can promote unwanted chemical changes. We've seen how sunlight exposure dulls product, invites degradation, and leaves batches with off-odors that customers notice right away. Simple shade, or housing in a warehouse with restricted light, keeps this sort of material much closer to its intended form.
Heat can be subtle. Warehouses sometimes spike in temperature in midsummer, or near steam lines. That bit of extra warmth hastens evaporation and can foster flammable vapors—unwelcome in any chemical workplace. Industry best practice keeps methyl o-benzoylbenzoate in cool, well-ventilated rooms, away from any ignition source. We physically separate storage areas from production lines and any space with welding or grinding equipment. It’s rarely as simple as “store in a cool place”—we check actual temperatures, track seasonal changes, and monitor areas for overheating before ever scheduling a drum delivery.
Humidity and Container Integrity
Chemical purity starts with packaging. In our plant, product leaves sealed in high-quality containers—steel drums with secure gaskets or polyethylene-lined barrels. We stick with these choices out of sheer experience. Moisture sneaks in around loose caps or dented barrels, causing hydrolysis or contamination. Routine inspection makes a difference; we pull aside containers with any sign of rust, swelling, or chemical staining. Once opened, partial drums often attract moisture from humid air, so our team works quickly, draws off needed quantities, and reseals with dry nitrogen when needed for sensitive runs.
Fire Hazards and Shared Warehouse Space
Aromatic esters like methyl o-benzoylbenzoate fall into moderate hazard classes due to their flammable nature. Real life shows that warehouse fires can start small—static discharge, faulty wiring, careless smoking—then move through rows of drums in minutes. We separate this class of material from strong oxidizers, acids, and bases. There’s no place for shared storage with sodium chlorate or perchloric acid. In joint facilities, we review what’s on the next pallet—keeping compatibility matched, not just for insurance, but to keep our own batches pure and our neighbors safe.
Employee Training and Labeling
No storage plan succeeds without people. We hold regular briefings on drum handling, spill response, and correct PPE. Labels never just name the material—they flag hazards in plain language. Overflows have happened; someone without training moves a drum incorrectly or fails to spot a leaking seal. With training in place and clear labels present, mistakes become rare, and incidents get contained fast.
Solid storage strategies build trust—from regulatory inspectors who count on compliance, to customers who need pure, reliable chemistry. Knowledge from the floor shapes every standard, always rooted in daily experience with materials like methyl o-benzoylbenzoate.
Purity Specification Defined by Real-World Experience
Every batch of methyl o-benzoylbenzoate produced in our plant tells the story of discipline in chemistry. Over years of manufacturing, purity doesn’t just get attention because a certificate requires it. In practice, the ideal figure stands above 99%, and most technical conversations settle around 99.5%. Several industries won’t accept less.
A lab instrument might flag 99.3% or 99.7% for a finished lot. Chemically, that minor swing can matter. In photoinitiators, coatings, or pharmaceutical intermediates, a half percent gap leaves a window for side reactions or colored by-products. For us as producers, that means consistent monitoring, not just an afterthought in a final QC check.
What Drives the Demands on Purity?
The applications set the standard. Paint chemists and engineers turn away from shipments with detectable benzophenone or unreacted acid. These aren’t scare stories—customers share instrumental chromatograms highlighting any trace that doesn’t belong. Any single spike brings a call and the lengthy process of problem investigation.
By the time a batch leaves our warehouse, we’ve tracked purity at multiple points. Raw material checks start things off, as methyl benzoate or o-benzoylbenzoic acid come with their own impurity risks. Throughout esterification, we sample and run thin layer chromatography, check clarity, and pull for HPLC analysis. As the final product cools, trace crystals or off-odors point to incomplete reaction or side products—both meaning internal rejection, not customer complaints later.
Technical Challenges Keeping Purity High
One risk comes from overheating; reaction at excessive temperature might create transesterification by-products. Tight pH control prevents hydrolysis or acid-catalyzed side paths. Solvent choice, reaction time, and full washing after synthesis each play a part. Solvents that seem purely functional in theory often introduce their own impurities. That’s why a closed-loop, batch-controlled system makes the difference.
Operators in the control room don’t just watch screens—they pull actual material, check for cloudiness or crystals against proper samples, and feel accountable to their coworkers, not just to paperwork. Fresh filters stop particulate contamination. We’ve improved yields and purity by adopting finer filters and more accurate dosers for the reagents.
What Happens When Purity Drops?
If a lot comes in at 97% or 98%, even with a passable appearance and main elements, it gets flagged internally. It’s a big deal. Blended or reprocessed lots won’t impress a regular customer. Each impurity brings performance risks—unpleasant smells, color instability, unpredictable photoinitiation. Clients who manufacture inks, resins, and coatings measure everything. Their standards don’t flex, and rightly so.
How Specification Connects to Trust
After years on the manufacturing floor and in meetings with customers, the firm truth is that a posted purity number isn’t just chemistry—it’s a promise. Real people in R&D and production rely on it, as do product managers who’ve faced costly recalls when a sub-spec drum slipped through. For our part, achieving and maintaining >99.5% purity isn’t only about detection limits or statistical averages. It’s each person in the plant realizing every shipment is an extension of our own standards.