Methylisothiazolinone: An Editorial Perspective from a Chemical Manufacturer
Historical Development
Developing methylisothiazolinone (MIT) into a mainstream preservative took a bit of persistence through the late 20th century. We saw its emergence decades ago during a period when industry demanded cost-effective biocides that handled a broad spectrum of bacteria and fungi. Early on, researchers noticed that combining efficient microbial control with favorable water solubility could address issues plaguing everything from paints to personal care. R&D teams refined isolation processes and lab-scale production in response to mounting microbial resistance to earlier preservatives. These efforts formed the backbone for moving MIT to full-scale industrial synthesis, supporting stricter microbial requirements in sensitive applications like cosmetics and household cleaners. Regulations changed, forcing everyone to innovate in order to meet performance needs while dealing with growing safety scrutiny.
Product Overview
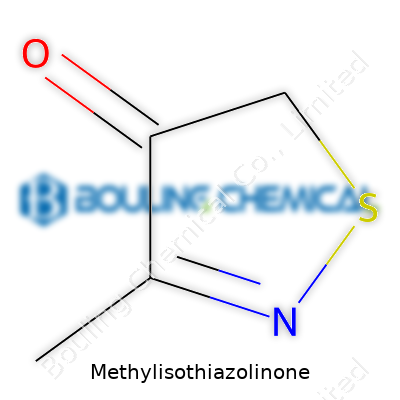
Methylisothiazolinone belongs to the family of isothiazolinones, which disrupt microbial protein synthesis. The molecule’s main appeal comes down to reliable broad-spectrum activity and easy compatibility with water-based formulations. We manufacture it as a pale yellow to colorless liquid, often supplied in dilute aqueous solutions to match downstream dosing practices across sector boundaries. MIT’s effectiveness at low concentrations in neutral to slightly alkaline pH mediums explains its widespread adoption in paints, coatings, adhesives, and shampoos. Raw material quality, process consistency, and minimal byproduct content make the real difference for end users who expect robust shelf life extension even in tough storage conditions.
Physical & Chemical Properties
MIT’s molecular formula is C4H5NOS, and its modest molecular weight gives it favorable handling characteristics during formulation. With a boiling point that allows stable storage in common warehouse environments, and a decent shelf stability under ambient conditions, MIT fits many applications. It dissolves readily in water and most polar solvents, an edge over traditional hydrophobic preservatives. Its sharp but faint odor identifies it in the plant, and we note the tendency to degrade with prolonged exposure to UV or strong alkaline media—details that must be considered during both manufacture and application.
Technical Specifications & Labeling
Manufacturers must guarantee purity levels as regulatory standards get tougher. We track active ingredient content down to parts per million, managing batch certification against ISO, REACH, and the relevant national chemical standards. Accurate labeling remains a non-negotiable, with hazard symbols, precaution statements, and expiry dating always featured. For MIT, labeling rigor covers more than compliance: it shapes market confidence, lays out safe transport guidelines, and underscores the commitment to traceability and recall efficiency in case of downstream issues.
Preparation Method
We synthesize MIT through the cyclization of appropriate thioureas with halogenating agents. The process involves precise temperature and pH management to ensure the selective formation of the isothiazolinone ring, minimizing formation of unwanted byproducts such as dichloro derivatives. Much effort goes into continuously recycling solvents, scrubbing vent streams, and controlling the effluent to meet environmental discharge norms. Batch size flexibility allows us to serve formulations ranging from small premium cosmetic lines to high-volume paint manufacturing. Process improvements in catalyst selection and continuous reaction monitoring have trimmed waste and improved cost efficiency over the years.
Chemical Reactions & Modifications
Chemists in the plant experiment with MIT in combination with other isothiazolinones or phenolic preservatives to reinforce spectrum activity and prolong microorganism knockdown. The molecule accepts minor structural tweaks, such as halogenation, that result in products with enhanced activity under different formulation pH conditions or storage temperatures. Our labs have run controlled tests to monitor the reaction profile when MIT is blended into high-reactivity pigment dispersions—a lesson in stabilizer compatibility and color fastness. Side reactions with certain surfactants or reducing agents have informed our dosing protocols and product advisory notes.
Synonyms & Product Names
Throughout the sector, MIT appears in documentation as 2-methyl-4-isothiazolin-3-one, MI, and several trade names depending on downstream applications. Paints and coatings may list it as part of “in-can preservative” systems, while home and personal care blends call it by international nomenclature, referencing INCI listings. Such synonym use by customers sometimes demands extra documentation and cross-referencing to avoid confusion during plant audits and safety assessments.
Safety & Operational Standards
Worker and end-user safety dictates every step from raw material storage to final application. Operators wear full PPE and follow rigorous hygiene protocols, particularly since regulatory agencies have zero tolerance for skin sensitization or improper airborne concentrations. Automated feed and closed transfer systems minimize exposure, and real-time monitoring flags any process drifts that could raise occupational health risks. Each plant adheres to regular trace emissions audits and maintains strict records for review during licensing renewals and routine inspections.
Application Area
MIT found its way into paints, glues, liquid soaps, and personal hygiene products, carving out a role as an all-round biocidal preservative. In the coating sector, MIT preserves water-based formulations in cans for extended shelf life, preventing microbial sludge buildup which can clog machinery and spoil entire production runs. Personal care producers favor MIT for its compatibility with surfactant-rich bases and its minimal impact on product color or fragrance. Industrial and institutional cleaning formulators integrate MIT into their blends to guarantee microbial control down to the last drop, even under demanding storage conditions.
Research & Development
Our R&D teams collaborate globally, seeking ways to boost MIT’s performance while reducing skin sensitization risks. We perform micro-scale challenge tests on new formulations, exposing them to real-world contaminants picked up from user handling and storage. Innovations include encapsulation technologies and slow-release systems, which lower exposure peaks and shrink allergenicity potential. Studies run every year to determine minimum effective dose in live application scenarios—not just in laboratory glassware. Ongoing work examines synergistic effects with newer antimicrobials and nanomaterial additives, aiming to stretch preservative performance without breaching legal use limits.
Toxicity Research
Toxicological scrutiny on MIT intensified as medical journals linked it to skin sensitivity, allergic dermatitis, and other contact reactions. Manufacturing plants follow the evolving literature closely, designing occupational safety regimes around latest exposure data. Even minute concentrations—measured in parts per million—attracted clinical investigation, driving us to collaborate with dermatologists, toxicologists, and regulators. Detailed patch testing and long-term dermal studies have clarified how modified formulations can retain biocidal benefits while cutting the risk profile for end users and plant workers alike. Efforts to develop robust in vitro alternatives also gain momentum, promising safer, more predictive evaluation before field trials.
Future Prospects
Global demand for reliable, non-antibiotic biocides continues to push MIT research and manufacturing forward. Regulatory tightening raises the bar on allowable limits, toxicity thresholds, and labeling demands. Our forecast expects stricter EU and Asia-Pacific controls, with the U.S. catching up as science drives regulatory policymaking. As users want less allergenic and more environmentally friendly formulas, industry pivots to develop hybrid preservative systems, green chemistry production routes, and next-generation stabilizer blends that outperform single-use MIT. Sustainable sourcing, energy-efficient processes, and recyclable packaging have turned from optional add-ons to core production priorities—transformations that echo up and down the value chain, influencing not only formulation science but public trust and market access. New entrants in isothiazolinone chemistry might eventually displace MIT, but for now, steady investment in operational quality and cutting-edge safety research remains the best path to responsible production and application.
Practical Experience with Methylisothiazolinone
Methylisothiazolinone, or MIT as it’s often called in the factory, shows up early on the ingredient list for a good reason. Most of us don’t give much thought to what keeps paints, detergents, and many personal care products from turning foul during storage. The enemy here is microbial growth. Water-based formulations attract bacteria and mold the way sugar attracts ants. Methylisothiazolinone stops that before it can start.
Our plant engineers watch for contamination in the production lines. Left unchecked, even a small colony of bacteria can ruin an entire batch—losses aren’t just financial; we’re talking about the cost to reputation, customer trust, and the stress on staff. By adding MIT, we cut down on spoilage almost to zero. It saves time, slashes waste, and lets our team focus on improving quality, not crisis management.
Why We Rely on It
We trust MIT because it gets results at low concentrations. Just a fraction of a percent matches up against the kind of aggressive yeast or bacteria that sneak into big mixing tanks. Preservatives that demand higher doses often shift product smell or texture. In today’s markets, no customer accepts odd odors in liquid soaps or sticky residues in wall paints. MIT keeps things fresh without changing the end product—a make-or-break difference in our line of work.
Regulations across North America and Europe push us to test every batch. Overuse of any preservative draws heavy scrutiny, and customer sensitivities matter. Our lab runs patch testing of finished goods for skin tolerance before shipment. The team pays close attention to scientific research, especially studies about allergies. The industry made changes—lowering max levels and swapping out formulations for certain products—because it matters to us that what we send out stacks up to safety standards and scientific knowledge.
Challenges and Improved Solutions
MIT’s not without controversy. Some customers report skin reactions, especially in leave-on cosmetics. These claims aren’t just stories; our R&D staff attends dermatology conferences and reviews clinical trials. The European Union banned MIT in leave-on items like face creams, and our safety team changed recipes as a result. For rinse-off products—think shampoos, paints, cleaners—it remains useful. We use advanced monitoring to measure down to parts per million. The plant invests in closed-system pumps to limit worker exposure during handling.
Finding substitutes takes work. Natural preservatives lack the punch of MIT or create their own risks with allergens. Some brands want “preservative-free” printed on the label, but waterborne products demand some form of protection unless customers enjoy frequent recalls and shelf spoilage. Continuous dialogue with raw material suppliers helps us stay on top of new developments. Keeping an open channel with our buyers, including listening to allergist feedback, ensures we never lose sight of the people using our products—not just engineers in the production hall.
The Road Ahead
MIT continues to fill an important gap in manufacturing for now. Every change in regulation or consumer trend brings fresh challenges. Our company pushes for cleaner alternatives but recognizes the risks of hasty replacements. Careful testing, transparency on ingredient choices, and a strong focus on product quality keep our business viable. We see MIT not as a shortcut, but as the baseline standard for safeguarding goods in a responsible way, for now and into the future.
Direct Experience in Formulation
Every day, our chemists weigh choices for ingredients that balance protection of products and safety for the people who use them. Methylisothiazolinone, often called MIT in our labs, finds use as a preservative in a wide range of liquid personal care and household products. It goes into shampoos, conditioners, liquid soaps, paints, and cleaners for a good reason: it keeps microscopic threats like bacteria and fungi from taking over. Many water-based products grow bacteria faster than most realize, so without robust preservatives, simple daily use can turn a safe formula into a petri dish.
The Challenge of Sensitization
Science has a clear message about methylisothiazolinone and its family of isothiazolinones—use it carefully. Reports from dermatologists and hospitals have increased in recent years, especially across Europe, pointing out rising cases of allergic contact dermatitis linked to this ingredient. Rarely does a week pass without a case where skin reddens, blisters, or itches in response. Machines in our plant process tiny amounts, but even trace exposure becomes a concern as more people develop sensitivities.
Respecting Regulations and Limits
No manufacturer can ignore the decisions made by global authorities. The European Union banned the use of MIT in leave-on products. For rinse-off products, only very small quantities are allowed. The United States still permits use in both, but there is ongoing discussion about lowering the allowable concentration. Realistically, more countries will continue to tighten their stance as new data emerges.
Balancing Preservation and Risk
Dropping MIT entirely from production lines has ripple effects. Plant operators see costs rise with shorter shelf lives or substitute preservatives that affect texture, odor, or performance. People expect smooth, pleasant products, and many alternatives require larger amounts or do a less thorough job protecting formulas. This increases the odds a product could spoil before someone finishes the bottle. That said, health always takes priority. We see evidence that reduces MIT alongside other isothiazolinones lowers cases of allergic reactions in the general population. Making MIT-free products is now common in categories intended for babies and those with eczema.
Continuous Testing and Industry Responsibility
Routine patch testing plays a large role in checking for safe levels. Our labs run assessments regularly, looking for signs that a formula could trigger irritation. These tests are not just about legal compliance—they offer real-world safety data. Working with raw MIT requires strict procedures and personal protection at the industrial scale, further evidence that respect and vigilance are critical.
Moving Toward Safer Alternatives
Every new ingredient goes through the same scrutiny as MIT did when it first hit the market several decades ago. Today, rising consumer preference for “free-from” labels pushes our team to test new blends of preservatives. Some latest-generation alternatives come with their own challenges, but ongoing investment in R&D promises safer products and more reliable shelf life for years to come.
In Summary
Real-world experience confirms MIT remains effective against microbial contamination, but risk of skin sensitization means manufacturers carry a responsibility to minimize use, follow science-based limits, and seek safer replacements where possible. The safest product is only ever possible with constant vigilance, listening to both regulators and the people who use what we make.
Why Manufacturers Use Methylisothiazolinone
As a chemical manufacturer, we know the reasons methylisothiazolinone ends up in so many consumer and industrial products start with its strength as a preservative. Our labs have put this molecule through rigorous stability and microbiological challenge testing. Bacteria and molds multiply where water lingers, such as in shampoos, lotions, paints, and household cleaners. One small dose of methylisothiazolinone knocks down unwanted growth fast, extending product shelf life and guarding against product spoilage that can cost brands money and reputation.
Common Products Containing This Chemical
Try looking around the bathroom or under your kitchen sink. You’ll likely spot methylisothiazolinone in liquid hand soaps, shower gels, and body washes—especially the “clear” types, not soap bars or powders. Baby wipes, makeup removers, and some moisturizers sometimes rely on it too, mainly in water-based formulas. Bottles of dishwashing liquid and all-purpose spray cleaner show this ingredient on their labels, since they contain enough water to tempt microbial growth without chemical protection.
Our customers that make paints and coatings are some of the most frequent buyers. Waterborne wall paints and varnishes spoil quickly once exposed to air, particularly if kept in a humid garage or basement. Surface disinfectants and air fresheners offer another use; without preservatives, open spray bottles could breed bacteria inside just a few days. The preservative lends a longer time frame between filling, shipping, and actual use, especially in warmer climates or warehouses.
Complexity of Safe Use and Clear Labeling
As much as manufacturers value methylisothiazolinone's action, the safety discussion deserves as much attention as its technical benefits. We addressed queries from the cosmetics and personal care industry for years as dermatology research began highlighting skin sensitivity issues. Now, both Europe and North America have shifted focus to minimize or eliminate this preservative from rinse-off products like hair care, and have set tight limits for leave-on applications such as lotions.
We field regular updates on allergy rates and compare those numbers with the raw data from production and customer feedback. This dialogue with regulators, medical professionals, and end users has seen our technical teams offer alternative blends or carefully control manufacturing environments to reduce cross-contamination. We also supply technical documents—far from sales brochures—to help our partners understand risk management and compliance strategies, aligning with new legal limits as soon as enforcement rolls out.
How Industry Approaches Alternatives
Shifting away from methylisothiazolinone across various applications calls for significant investment and collaboration. The search for equally effective, stable, and cost-competitive substitutes has led to lengthy development cycles. Microbial challenge tests and real-life shelf life trials help screen new candidates. Our product development team keeps sustainability and low skin sensitization profiles at the core of new preservative blends, aiming for transparency in formulas supplied to consumer brands.
Supporting downstream partners through reformulation, technical guidance, and education remains a daily priority. Our understanding—gained from producing and handling these chemicals ourselves—pushes us to focus on safer chemistry. This ongoing process ultimately helps manufacturers, retailers, and consumers stay informed about what preservatives go into common household products, how regulations shift over time, and why transparency in labeling and production remains so important.
Understanding Customer Concerns
People often ask about the risk of allergies from methylisothiazolinone. This question comes up for good reason. The ingredient works well as a preservative and gives products a longer shelf life. At the same time, stories about skin reactions tend to get attention—especially in personal care or cleaning goods. Working directly with this chemical every day and talking to customers in production lines, I've seen the real-world impact that allergy risk creates.
Lessons Learned in Daily Chemical Manufacturing
Anyone operating reactors or filling drums gets close to the process and understands the precautions inside the plant. Direct skin contact with methylisothiazolinone feels unpleasant, even in small amounts. Our lab crew uses gloves, ventilation, and washing facilities for that reason. It’s common knowledge among line workers and supervisors. Years ago, dermatitis cases increased among operators before the implementation of new safety steps.
Workers aren’t the only ones at risk. End users—the hair stylists, janitors, household customers—may see contact dermatitis. In Europe, regulators documented a rise in allergic reactions from leave-on cosmetics that used this ingredient. Many manufacturers have since responded by lowering concentration, switching to rinse-off formulations, or moving to blends with other, milder preservatives.
Guidelines Shape Real Solutions
Some ingredients can barely be replaced, but guidance from regulatory bodies sets the direction forward. For instance, the European Union and other markets restrict or ban methylisothiazolinone in certain formulations. Industry sets limits below these thresholds, often through testing and pilot batches. Our technical team works with big consumer brands to reformulate. Often, we test preservative levels starting at the lowest percentage possible. Any new formula that reduces the risk of allergies gets fast-tracked.
Routine patch testing with volunteer panels shows where risk levels stand. No manufacturing process is perfect; the need to track skin reactions never fades. Technical support staff record these results and relay findings back to our chemical engineering crews. The direct feedback loop improves both raw ingredient quality and finished product safety.
Balancing Protection and Safety
Preservatives do an important job: stopping mold, bacteria, and spoilage. Yet experience shows that methodical control and clear communication beat simply increasing doses. Fixing a single preservation problem by raising chemical concentration may open the door to skin reactions. Cleaner production and alternative blends reduce pressure on a single ingredient and keep customer satisfaction up.
Clear labeling on drums, shipments, and product batches quickly informs customers. Safety data sheets cover medical guidance for exposure. Our team keeps up with reports from dermatology clinics and regulatory alerts. These steps limit the number of unexpected reactions and help protect those in frequent contact with our chemicals.
Pushing for Safer Choices
Incremental changes in chemical manufacturing often come from practical experience. Equipment upgrades, updated plant training, and ongoing consultation with clients keep methylisothiazolinone in check. Back on the plant floor, every improvement boosts worker health and makes the entire supply chain safer for customers. In the end, decisions about ingredients such as methylisothiazolinone work best when supported by open discussion and a steady stream of real-life data.
Stepping into the production line, I see methylisothiazolinone as more than a name on a drum. This preservative has powered the safety of shampoos, lotions, and personal care products for decades. It stops bacteria and mold from spoiling formulas long before a retailer places them on a shelf. From a chemical manufacturer’s standpoint, it delivers reliable, tried-and-tested performance for broad-spectrum preservation. Chemical producers like us carry a responsibility to follow the science and regulatory evolution around everything we deliver.
Shifts in Regulations Across Markets
Regions have taken different paths on methylisothiazolinone. In the European Union, the ban hit hard. As of 2017, using methylisothiazolinone in leave-on cosmetics—think creams and sunscreens—is prohibited. For rinse-off products like shampoos and body washes, there’s a maximum allowed concentration, far lower than before. Canada and Australia track close to these limits. The United States, for now, maintains fewer restrictions, but we notice a shift in manufacturer formulation as pressure grows from advocacy groups and retailers wary of allergens.
Allergic reactions and growing sensitivity stories in the press changed everything. Scientific committees in Europe reviewed the evidence and flagged contact allergies at rising rates, particularly among hairdressers and consumers using products frequently. The regulatory decisions didn’t come from speculation—they reflected thousands of real-world case reports. For any chemical manufacturer, allergy-related concerns shape how we manage hazards, talk with customers, and design next-generation preservatives.
Why This Really Matters
Regulatory changes ripple straight into how manufacturers operate. One day, a customer calls and asks about the new law in France; the next, an exporter inquires how our ingredient will fare at customs in Brazil. These questions turn into urgent reformulating projects. Labs work late to swap out methylisothiazolinone for alternatives, but swaps pose chemistry puzzles—think stability, shelf life, and cost. Each new material comes with its own safety data, its own environmental impact profile, and its own learning curve in production equipment. Raw material prices shift as demand for alternatives grows. Sourcing sometimes moves across continents. These are not theoretical headaches—they are daily challenges for the people who make the stuff from scratch.
Consumers often see “no MI” or “MI-free” on bottles and think the issue is solved. From the lab floor, rarely do we get “free” results at the same level of performance or economy without months of trials and failed batches. Some new preservatives are less effective at low doses or need higher use levels, increasing costs. Trends swing: years ago, parabens were the villain; now, manufacturers juggle multiple bans and watch for emerging issues on newer molecules.
Where Do We Go From Here?
One solution lies in open collaboration between regulatory agencies, chemical producers, and finished-goods manufacturers. When agencies publish clear guidance, our researchers can focus. Sharing more real-world data on skin sensitization helps target ingredients for reformulation. Investment continues into biotech preservative development, drawn straight from nature’s own solutions to microbial contamination. Already, my team has pilot projects with plant-derived antimicrobials and probiotic ferments. Results show promise, but scaling these up for hundreds of tons per year brings fresh engineering challenges.
The methylisothiazolinone story keeps evolving, shaped by allergy science, consumer demand, and logistics realities. Manufacturers have to adapt quickly, with a steady focus on safety and transparency from the supply chain forward. In the end, producing safe, stable, and accessible products always means constant change in the lab and the factory—and we owe that to everyone who trusts what comes out of our doors.