Sodium Ethylparaben: A Manufacturer’s Perspective
Historical Development
In the chemical industry, the story of parabens begins nearly a century ago, when researchers searched for reliable preservatives for cosmetics and pharmaceuticals. They understood spoilage didn't just waste resources—it threatened health. Ethylparaben stood out early for its broad-spectrum antimicrobial activity and its gentle profile when compared to stronger synthetic agents of the day. Sodium ethylparaben, a water-soluble salt of ethylparaben, entered the scene as formulators in the 20th century demanded easier integration into aqueous systems. The story tells of advances led not just by corporate innovation, but by practical challenges faced daily in compounding rooms and production halls. Through hands-on encounters, improvements followed: higher purity production, more consistent supply, and careful scaling that addressed both regulatory and consumer scrutiny.
Product Overview
Our plant produces sodium ethylparaben to support food preservation, cosmetics, and pharmaceuticals. We focus on high quality, known for low impurity levels and predictable performance in both large- and small-scale applications. The salt brings enhanced solubility and stability, which practical users value because batch losses trace back to issues like precipitation or incomplete mixing. By preparing sodium ethylparaben through precise chemical conversion, we gain control over critical grade parameters, supporting clients who demand traceability and repeat results.
Physical & Chemical Properties
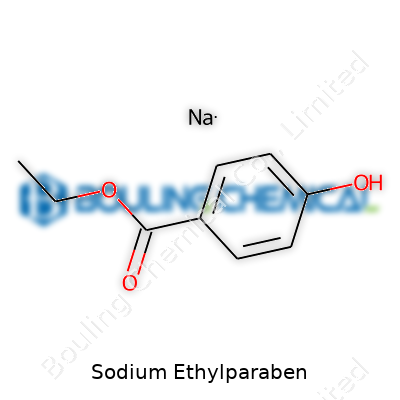
Sodium ethylparaben appears as a white, odorless powder or fine-granule solid. Its structure delivers benefits for water-based product designers; it dissolves swiftly and stays clear at common formulation temperatures. Chemically, the molecule consists of the sodium salt of a para-hydroxybenzoic acid ethyl ester, meaning we see both the paraben core’s well-documented functionality and the sodium group’s ability to enhance solubility. The melting point sits comfortably above typical climatic ranges, so storage and transport avoid the headaches of caking or melting. This stability allows end users to focus on the fine points of their recipes rather than troubleshooting preservative performance.
Technical Specifications & Labeling
Industry standards for sodium ethylparaben are strict, reflecting its use in sensitive environments. Manufacturers validate each lot with tests for purity—often above 99%—and for common impurities, especially related phenolic compounds or residual solvents. We maintain lab archives for every batch, as regulations occasionally require retrospective audits. Labeling leaves no room for ambiguity; end users need to know the net weight, batch, production date, and shelf life. Even the way packages are sealed or the style of tamper-evidence practices developed with input directly from major users, who have faced losses and regulatory fines from less diligent suppliers.
Preparation Method
From a process perspective, preparing sodium ethylparaben involves neutralizing ethylparaben with a stoichiometric amount of sodium hydroxide in a controlled aqueous medium. We monitor temperature and pH tightly to avoid hydrolysis or side reactions. Unwanted by-products can create downstream problems, not just in analytical tests but in final product behavior such as color, odor, or unintended reactions with active formulation components. The precipitated salt is filtered and dried using methods refined over years of experience. Production teams work directly with our R&D scientists, providing feedback that leads to subtle tweaks in crystallization rates and drying protocols.
Chemical Reactions & Modifications
Sodium ethylparaben maintains stability under a variety of pH and thermal conditions commonly encountered in product manufacturing. In some processes, this salt undergoes further transformation to yield custom preservatives, or interacts with other stabilizers, surfactants, or chelating agents. Runaway saponification remains rare but does occur, especially in highly alkaline environments. Modifications, including ester group swaps or integration into polymeric systems, open a path to more specialized preservatives where unique textures, release rates, or compatibility features are called for. These adjustments reflect years of listening to chemists working directly at the bench, troubleshooting or developing new products—not just copying academic literature.
Synonyms & Product Names
Across regulatory filings and supplier catalogs worldwide, sodium ethylparaben travels under a breadth of synonyms: sodium 4-(ethoxycarbonyl)phenoxide, sodium ethyl p-hydroxybenzoate, and CAS numbers in pharma-grade documents. Standardization in naming never keeps pace with everyday usage, and we field regular questions from buyers who face regulatory agencies demanding reconciliation of paperwork and labeling. Our technical support team receives frequent requests to clarify synonym relationships—a task best tackled by those who produce and certify the actual product.
Safety & Operational Standards
Handling sodium ethylparaben in a manufacturing setting means respecting its power as both a tool and a chemical. Safety data guides our approach: exposure controls, protective clothing, and ventilation to minimize dust. We maintain strict training for both seasoned technicians and new hires. Spills find themselves neutralized rapidly and waste controls address both environmental and regulatory standards. More than checklists, these routines grow from incidents, near misses, and the frank sharing of operational experience among our team and trusted partners.
Application Area
Formulators use sodium ethylparaben to inhibit microbial growth and spoilage in creams, lotions, shampoos, and liquid pharmaceuticals. In food, its role is more tightly regulated, but it still finds niche applications for select categories thanks to its rapid dissolution and lack of taste impact at permitted levels. Multi-use plants—those that switch from batch to batch—choose this salt for quick changeovers and ease of cleaning, not to mention the historical record of consumer tolerance and safety. We see direct feedback at technical conferences, where professionals share both successes and pain points working this preservative into demanding new product lines.
Research & Development
Ongoing research centers around higher-purity grades and blends that extend shelf life or improve compatibility with emerging actives in cosmetics and drugs. Analytical teams evaluate trace-level contaminants, seeking to stay ahead of shifting regulatory limits. Bioavailability and controlled release remain active research paths, often with university and contract labs feeding data directly into our pilot-scale process improvements. Waste reduction, energy optimization, and process safety studies roll out from our own journals, and by working in collaboration with research partners, new test methods and application fields keep sodium ethylparaben near the top of the preservative toolbox.
Toxicity Research
Regulatory scrutiny has driven extensive studies into the toxicity and allergenic potential of sodium ethylparaben for decades. Acute and chronic exposure experiments form the backbone of both our internal audits and submissions to approval agencies. Endocrine disruption takes center stage, with detailed literature reviews and original research continually redefining limits and labeling requirements. Physicians and toxicologists regularly consult us about metabolite behaviors, absorption rates, and elimination statistics. Few topics generate more spirited debate at national and international regulatory gatherings, leading to ongoing evaluations and, at times, reformulation projects.
Future Prospects
The role of preservatives continues to evolve, driven by consumer demand and regulatory oversight. We see a future where sodium ethylparaben coexists with newer, greener alternatives, not replaced outright but used in smarter, lower-exposure applications through combination strategies. Automation and AI-based monitoring help us improve batch consistency and root out micro-level contaminants. As emerging markets demand more stringent quality controls and the pharmaceutical and personal care industries move toward transparency, manufacturers with a legacy of quality and adaptability are best positioned to answer these challenges. Our teams—working hands-on in production and with a finger on the regulatory pulse—will remain central to this ongoing evolution.
Where Sodium Ethylparaben Finds Its Place
Inside the plant, we work with Sodium Ethylparaben every week. This preservative brings dependable microbial protection to products people use every day. Unlike preservatives with broader acceptance issues, Sodium Ethylparaben offers a combination of effectiveness and stability. In our own lines, we see the demand coming directly from personal care, pharmaceuticals, and the food sector. Each of these industries brings its own expectations, but the fundamental need—protecting products from the growth of bacteria and fungi—remains the same.
Why Preservative Choice Matters
We talk directly with research teams who balance safety, shelf stability, and compliance. Sodium Ethylparaben stands out because it dissolves easily and integrates smoothly with water-based systems. Companies rely on this because the last thing anyone wants is a compromised batch that reaches the market with contamination risks. In creams, lotions, liquid medicines, and some processed foods, microbial contamination not only shortens shelf life—customers lose trust, regulatory bodies step in, and recalls become a risk. From our side, a preservative that delivers performance without complicating formulation means fewer headaches at every stage.
Consistency Through the Manufacturing Process
Our process must keep quality high and impurities low. We invest in monitoring every run, not just batch testing at the end. Every team member knows that even a slight impurity can compromise a batch or disrupt our clients’ product stability tests. Sodium Ethylparaben gives us a reliable profile; our teams measure stability across a range of pH levels and temperature conditions. This consistent behavior reduces troubleshooting down the line for manufacturers who rely on our technical support.
Meeting Regulatory Challenges
Regulatory expectations never stop evolving. Authorities scrutinize preservatives more now than in decades past, driving requests for transparency and documentation. From our conversations with clients, clarity on permitted usage levels and toxicology profiles remains essential. Sodium Ethylparaben is subject to limits in different markets—the US FDA, European Union food safety authorities, and others set these. We maintain up-to-date technical files and respond to audit requests swiftly to support our partners through each approval cycle.
Potential Solutions and the Road Ahead
As consumer demand shifts toward products with fewer preservatives, formulators look for actives that deliver safety while keeping ingredient lists recognizable. We engage with development labs experimenting with natural alternatives, but cost, efficacy, and long-term stability are tough hurdles. For now, Sodium Ethylparaben represents the balance point: a proven material, with decades of data, providing safety outcomes for a wide range of products. By continuing to refine our process and maintaining open lines of communication with customers, we can help them adapt to new regulations or customer preferences as they arise.
Everyday Impact
Sodium Ethylparaben rarely gets noticed by consumers; that’s a mark of success for a preservative. For us—right at the source—the job goes further than shipping a chemical. Our work ensures that the daily products on store shelves remain safe, reliable, and trustworthy from the moment they leave the factory until they reach homes around the world.
Experience Shaped by the Shop Floor
In the lab and on the production line, our relationship with Sodium Ethylparaben runs deep. We’ve seen the compound progress from raw ingredient to finished preservative—critical in many cosmetic and personal care products where controlling mold and bacteria matters. The importance of product safety rests not only in the science but in how well materials stand up to real-world usage. Years of testing, quality control, and feedback from seasoned formulators guide how we produce and judge this compound.
Scientific Consensus and Regulatory Review
Sodium Ethylparaben, a member of the paraben family, often draws questions due to ongoing debates about parabens as a group. Over the years, reputable organizations—including the U.S. Food and Drug Administration and the European Commission’s Scientific Committee on Consumer Safety—have published findings on parabens in cosmetics. Regulations cap maximum concentrations. For sodium ethylparaben, the limits sit far below what could cause acute or chronic health problems in humans.
We don’t cut corners with analytical purity or quality. Rigorous batch testing, both in-house and through accredited third parties, verifies specifications. This means picking off-target contaminants, watching for unapproved analogs, and confirming batch reproducibility. Not every synthetic preservative gets this degree of scrutiny—market pressure and strict regulations steer our every move.
Addressing Ongoing Concerns
Years ago, research suggested parabens might disrupt hormone function, but actual risk depends on exposure and absorption. In our line, formulas typically use minute concentrations—well within regulatory limits and below thresholds flagged in toxicology studies. Most sodium ethylparaben applied to skin does not penetrate deeply or accumulate; the body processes and excretes it efficiently. Human allergic reaction rates to this preservative hover at low levels. Our own tracking, as well as follow-up by downstream users, supports this.
Misunderstandings stem from generalizing studies or failing to contrast animal versus human exposures. Scientific advocacy groups sometimes highlight worst-case scenarios, but meaningful safety depends on real-world concentrations, consistent production quality, and transparent labeling. We stick to these principles in both research and scaling up for clients.
Pursuing Safer, Smarter Formulation
Market demand for “paraben-free” labeling hasn’t gone unnoticed. We routinely help formulators adapt, trialing alternative preservatives and sharing real stability data. The quest for less-sensitizing, biodegradable compounds is continuous, but not every substitute measures up to sodium ethylparaben’s microbial performance in all product types. Modern consumers expect shelf-life, safety, and transparency—none at the expense of product reliability.
Practical Solutions and Looking Forward
Instead of fueling standoffs around legacy compounds, we back investment in sound research and open communication. The standard for sodium ethylparaben has never been static. We stay close to ongoing studies, regulatory updates, and feedback from cosmetic chemists handling everyday safety concerns. Responsible chemistry means you weigh risks honestly while remaining grounded in measurable facts. Manufacturing for people means you never lose sight of either.
Over the decades, sodium ethylparaben has become a familiar ingredient on both ingredient lists and chemical order sheets. We handle metric tons of it in our plant every year, and its role as a preservative is tried and tested. Formulators rely on its ability to keep products shelf-stable, especially across the food, pharmaceutical, and personal care sectors. But recent news headlines have brought attention to the possible side effects connected with its use, and I want to share our perspective as a manufacturer deeply familiar with its production and behavior.
Known Effects: Skin and Allergic Reactions
In day-to-day work at the plant, we never ignore the possibility of skin or eye irritation. Direct contact with the powder can lead to redness, itching, or mild inflammation. These responses show up mostly during process interruptions or cleaning. Typical protective equipment like gloves, goggles, and proper ventilation keep incidents rare. Some workers, especially those with existing sensitivities, can show symptoms even with low-level exposures. This matches documented data from toxicology studies: certain people develop contact dermatitis from repeated or prolonged exposure.
Final product manufacturers sometimes report cases of mild allergic reactions in end-users. Symptoms tend to include local redness, itching, or occasionally hives where the product was applied. Most users show no reaction; the ones who do tend to have a history of allergies to parabens or other preservatives. Still, it’s serious enough that we urge partners to assess formulation levels, especially in products meant for infants or those with sensitive skin.
Long-Term Exposure and Controversy
In our own plant health surveillance, we see no link between sodium ethylparaben and cancer or hormone disruption, either among our crew or through peer-reviewed health surveys. Scientific reviews do not classify sodium ethylparaben as a proven carcinogen or endocrine disruptor. Yet, uncertainty lingers among consumers, mostly due to debates about whether parabens can mimic estrogen in the body. Regulatory agencies in major markets like the EU and US continue to review research and have set strict limits to keep exposure well below any threshold of concern.
We track all relevant legal requirements and follow any new instructions closely. High purity processing, clear labeling, and strict batch analysis keep our output within allowable limits. Any material outside specification is held and never reaches customers.
Mitigating Risks: Manufacturer’s Tools
Risk management in the factory comes down to control and communication. We minimize dust, enforce personal protective equipment rules, and monitor the air for any build-up of fine particles. Employees are encouraged to report symptoms, and product safety data is up-to-date and accessible.
For our customers—formulators, product developers, and end-users—there’s value in clear dialogue. Choosing the right preservative level, understanding user profiles, and conducting robust patch tests reduce downstream reactions. We see the best results in collaborative product development, where real-world data and lab analysis feed back into safer formulations.
Always Evolving with the Science
Every piece of feedback and published research drives our improvement. The world keeps changing, and our responsibility is to adapt and deliver materials that meet both regulatory and real-world safety. Sodium ethylparaben remains a valuable tool, but using it wisely—supported by transparent sourcing, robust handling, and honest risk sharing—creates better outcomes for everyone down the line.
Every day, in our facility’s production halls, we work with a variety of preservatives. Sodium ethylparaben frequently comes up in discussions with clients who need reliable protection against microbial growth in their products. This ingredient, synthesized by our chemists under strict quality controls, delivers real performance in many finished formulations—be they cosmetic creams, lotions, or personal hygiene products.
Understanding Its Role in Product Integrity
The world of preservatives can appear crowded, yet not all choices hold up under scrutiny. Sodium ethylparaben distinguishes itself with strong antimicrobial action, especially against fungi and certain bacteria. We’ve run our own challenge tests, and the results have consistently shown a genuine reduction in contamination risks. The molecule’s effectiveness traces directly to its ability to disrupt cell membranes in microbes, keeping formulas stable as they pass through supply chains, travel on shelves, and end up in consumer bathrooms.
Our process control team watches for real-world situations that might reduce a formula’s shelf life, such as heat, humidity, and variable pH. Parabens, including sodium ethylparaben, have proven to be resilient in varied pH environments—from acidic to slightly alkaline—without losing their antimicrobial punch. We have noticed that water solubility provides extra convenience for manufacturers who struggle with stubborn ingredients that refuse to blend. The sodium salt dissolves easily, so our customers don’t run into undissolved particles or inconsistent preservation.
Weighing Safety and Regulatory Confidence
Safety shapes how we develop and recommend any preservative. Decades of study back up sodium ethylparaben’s safety profile at recommended levels. International regulatory agencies, after independent reviews of toxicological data, still allow its responsible use. We’ve reviewed studies examining absorption and potential hormone interactions. Practical daily exposures during typical product use fall far below any level of concern described in these reports.
Safety discussions in the media sometimes create confusion, but our customers often seek straight answers rather than emotional headlines. We share real, lab-based numbers drawn from validated studies rather than rumor. Our safety team reviews every batch, runs purity checks, and ensures compliance with all updated norms. If stricter limits ever emerge, we invest in plant upgrades and process improvements instead of shortcuts.
Addressing Current Industry Pressures
Preservatives face a tough crowd. Market trends favoring “minimalist” formulas create pressure on chemists to reduce or eliminate parabens. This movement challenges our R&D crew to develop new blends or optimize usage levels without compromising protection. We collaborate with customers to run side-by-side comparisons, documenting real contamination incidents before making any reduction.
Switching to alternative preservatives sometimes creates other problems: instability, harsh odor, or inadequate coverage. From our experience, sodium ethylparaben often outperforms these newer substitutes, especially when maintaining gentle skin feel and clear appearance are priorities. In honest bench trials, it provides broad-spectrum activity that helps extend product shelf life with minimal downside.
Looking Forward
Customers trust us to balance honest science and industry trends. We support more transparent labeling, which gives users the chance to learn where their preservatives come from and why they matter. Most importantly, we keep our labs open—to new information, improved synthesis techniques, and real-world feedback—so that our sodium ethylparaben and every other preservative we make stand up to daily use and scrutiny.
Looking at the Real-World Impact of Sodium Ethylparaben in Everyday Products
Every day in our facility, we handle and produce sodium ethylparaben for use in a wide range of cosmetics and personal care goods. Customers and partners often raise concerns about skin sensitivities and allergic reactions, so we pay close attention to real feedback from both the manufacturing floor and the end-users.
Sodium ethylparaben works well in a lot of products because of its reliable preservative properties. It can stop mold and bacteria growth, especially in water-based formulas like lotions and shampoos. Despite its benefits, some people raise questions about preservatives and whether they might trigger allergies or skin irritation. Our own manufacturing staff interacts with sodium ethylparaben almost daily—raw powder, dust in the air, liquid phases—giving us a unique window into potential health effects.
From our experience, routine industrial safety standards—like dust control, gloves, and goggles—take care of most workplace concerns involving direct contact with large amounts during processing. Direct skin contact with any powdered chemical can lead to dryness or slight irritation, often due to the physical properties—small particles absorb moisture from the skin, leading to a chalky residue. We rarely see persistent problems, and when they occur, workers mention relief after basic washing and moisturizing. The quantities and concentrations in consumer products are much lower than what raw material handlers face in manufacturing.
Clinical literature and published dermatology reports echo what we see: most people tolerate sodium ethylparaben well in cosmetics, far better compared to stronger preservatives or known allergens like fragrance mixtures, nickel, or formaldehyde releasers. The European Commission and US FDA allow sodium ethylparaben up to limits considered safe after skin safety reviews. Incidents of proven allergic contact dermatitis from sodium ethylparaben appear quite rare compared to other common cosmetic ingredients. Most dermatologists categorize it as a low-risk preservative.
In recent years, claims about "paraben allergies" have grown on social media and beauty forums. We regularly test our batches for impurities like free acids, residual solvents, and microbial contamination, since those can cause irritation. Sometimes a reaction may come from another component in the finished product, not the sodium ethylparaben itself. Well-controlled production, clear labeling, and ongoing consumer reporting keep risks low. On the rare occasions an allergy is proven, discontinuing use stops the problem.
Our responsibility includes listening closely to feedback and supporting further safety studies. Formulations for sensitive skin use minimal concentrations and undergo clinical patch testing before launch. People with a history of skin allergies should still consult with their dermatologist. For the large majority, using products with sodium ethylparaben poses a low risk, but vigilance stays important as consumer habits and product blends evolve.
Addressing Concerns Through Safe Manufacturing and Transparency
Long experience handling sodium ethylparaben teaches us that informed oversight, responsible production, and good communication with both customers and peer manufacturers go a long way to resolving questions about skin irritation and allergies. With accurate information and quality controls, manufacturers can match the genuine safety needs of the end user and support the health of the industry overall.