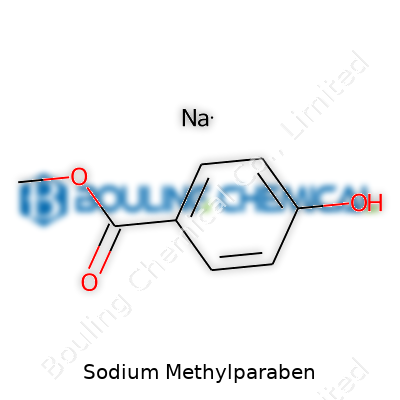
Sodium Methylparaben: An Industry Perspective
Historical Development
Parabens have shaped the history of personal care and pharmaceutical preservation. Sodium methylparaben, in particular, came into focus as formulators confronted microbial spoilage in aqueous products. Decades ago, chemists saw methylparaben’s potent antimicrobial effects and set out to address solubility challenges. This gave rise to sodium methylparaben—a sodium salt form that dissolved better in water, responded to demands for more stable and effective preservation, and reduced formulation headaches. This transformation mirrored shifts in manufacturing, where the need for longer shelf life and more stringent safety standards grew. Regulatory scrutiny, especially on product safety, pushed scientific progress. Researchers traced its metabolism and breakdown, ensuring it matched rising expectations for consumer safety. Sodium methylparaben didn’t just fill a technical gap; it set a new bar for preservative reliability as the world pushed for more robust solutions in everything from cosmetics to injectables.
Product Overview
Sodium methylparaben belongs to the paraben family of preservatives. This compound, the sodium salt of methylparaben, gained market share as a go-to ingredient for broad-spectrum protection against bacteria, yeast, and mold. Production scales up to industrial volumes to keep up with the demand. Its use stretches across several industries: from creams, lotions, and shampoos to syrups, injectables, and processed foods. Sodium methylparaben offers compatibility with wide pH ranges and prevents spoilage, minimizing recalls and waste. Decades of use have demonstrated its value, especially as formulators demand reliable preservation without triggering instability in delicate formulations. The salt form offers better solubility in aqueous solutions compared to the parent acid, opening more formulation options and supporting higher accuracy in process control.
Physical & Chemical Properties
Chemically, sodium methylparaben looks unassuming—white to almost white, crystalline powder or granules. A mild characteristic odor gives away its identity in bulk transfer rooms. It dissolves readily in cold water, which streamlines mixing during production runs and reduces risk of clumping. The taste in high concentrations turns slightly bitter, though use levels are low enough to avoid sensory issues in oral medications. The chemical formula C8H7NaO3 tells part of the story; it provides structural stability even under the challenge of repeated heating and cooling cycles. Melting points hover above regular handling temperatures, meaning there’s little risk of breakdown under normal storage. The molecule holds up in light and air exposure, lowering the burden on protective packaging. Over the years, we’ve logged minimal reactivity with common excipients, further reducing surprises during scale-ups or tech transfers to new process lines.
Technical Specifications & Labeling
Technical standards draw on pharmacopeial guidelines. Companies like ours assess appearance, assay purity by high-performance liquid chromatography, confirm identity through infrared spectra, and monitor microbial counts. Purity checks routinely give results above 99%. Heavy metal content and organic impurities are kept to the narrowest possible bands. Labeling aligns with both global and local regulations. For pharmaceuticals, labels report exact quantities per dose; for cosmetics, declaration follows ingredient lists mandated by local authority. At the manufacturing level, documentation tracks each step—from bulk synthesis to final packaging. Traceability keeps recalls rare and confidence high. Containers must protect from moisture, light, and cross-contamination; this demands not just tight seals, but robust secondary packaging to prevent accidental mixing with similar white powders common in manufacturing environments. Even the drum or bag material is selected to avoid plasticizer interaction over long-term storage.
Preparation Method
Our process begins with methylparaben. Reacting it with sodium hydroxide produces sodium methylparaben and water. The reaction proceeds in controlled vessels—temperature control, agitation rate, and pH monitoring matter to maximize conversion and prevent by-product formation. After the reaction, we filter and wash the solid to remove any unreacted alkali, then dry the product under reduced pressure. Particle sizing follows if a customer requires a specific mesh or flow property. Each lot is sampled and tested not only for chemical composition, but for solubility and stability. Years of process optimization help us reduce waste streams, capture reactants for re-use, and limit solvent exposure for operators. Data from each production run supports continual improvement; variances are tracked and root causes are addressed before the next batch ever starts.
Chemical Reactions & Modifications
Sodium methylparaben’s classic reaction—the hydrolysis of an ester—turns the methylparaben acid into a more water-soluble salt. Beyond this, the core structure tolerates various chemical environments, but doesn’t easily break down under normal use. Under severe acid or base, it can revert to methylparaben or further break down to p-hydroxybenzoic acid. Chemical modifications remain limited in commercial contexts, but recent research pursuits look at altering the alkyl chain or introducing ring substituents to target new preservation profiles. In the plant, we track stability during heat cycles and monitor pH in storage tanks to avoid unwanted hydrolysis or discoloration. The molecule’s predictability has helped keep unplanned downtime at bay.
Synonyms & Product Names
Sodium p-hydroxybenzoate, E219, and the sodium salt of methylparaben describe the same material on different continents. Some industries call it sodium methyl-p-hydroxybenzoate, and in code-heavy settings, E219 prevails. Trade names vary, but careful labeling on every drum and shipment clarifies the substance regardless of the supply chain language. Each synonym traces back to regulatory, academic, or trade preferences, but consistency across global markets reduces errors both in procurement and on the plant floor.
Safety & Operational Standards
Safety sits at the center of our operations. Although sodium methylparaben has a strong track record for low acute toxicity, we’ve built in layers of precaution. Bulk handling involves dust extraction and closed transfer lines; operators wear protective gear, especially during weighing and charging phases. Ventilation and spill containment plans reduce particulate risk in multi-product facilities. Wastewater monitoring ensures no uncontrolled release. Our documentation and training encompass all relevant handled volumes. We order periodic reviews as new research emerges—whether that’s a shift in cosmetics policy or new findings on trace impurities. Emergency drills rarely get used, but form part of audits that drive improvement and satisfy regulators who know that one slip can cost more than the value of any batch.
Application Area
Sodium methylparaben reach extends into high-standards industries first shaped by early batch failures and persistent contamination. In pharmaceuticals, it keeps both oral and parenteral solutions free from spoilage. Food producers rely on it for bakery, beverage, and ready-to-eat foods. Personal care remains the broadest adopter, pulling demand for everything from shampoos to serums and wipes. Water solubility overcomes barriers associated with other parabens, simplifying formulator’s decisions and reducing microbe-driven recalls. These advantages support innovation in product pipelines. Over the years—through reformulations and regulatory changes—the material has held its position in preserving safety and function.
Research & Development
Our R&D moves between incremental gains and big leaps. Teams refine synthesis to minimize residual solvents and process by-products. We partner with downstream users to monitor preservative performance in increasingly complex cosmetic matrices—multiphase systems, emulsions, and protein-rich foods throw up challenges in preservative efficacy. Work on analytical methods tracks ultra-low residue levels and stays ahead of evolving regulations. Toxicological studies, both in-house and through academic collaborations, yield detailed exposure profiles. As demand grows for preservative blends, researchers push for new combinations, lower usage levels, and synergies that maintain protection even as other formulation components shift. The drive for green chemistry keeps us experimenting with alternative raw materials and lower energy processing. Staying competitive often means anticipating regulatory moves and shifting scientific consensus—every R&D cycle validates the compound’s continued relevance through real application data.
Toxicity Research
Toxicity research forms a pillar of our stewardship. Pharmacokinetic studies show sodium methylparaben clears from the body rapidly, excreted mostly unchanged. Long-term rodent studies have probed for carcinogenic or endocrine effects, yielding no actionable risk at relevant use levels. Safety assessments regularly revisit exposure scenarios; each batch released conforms to restricted content rules for cosmetics and food. Our teams monitor findings on allergic reactions, particularly in populations with chronic exposure or sensitivity to multiple preservatives. In laboratory studies, we observe minimal cytotoxicity at product-representative concentrations, and occupational exposure limits rarely approach thresholds for concern. As pressure mounts from consumer advocates and regulatory agencies, we invest in ongoing data gathering, feeding results directly to both risk assessors and regulators. This feedback loop supports both regulatory confidence and responsible use in the industries reliant on sodium methylparaben.
Future Prospects
Market expectations point toward changes in both volume and regulatory burden. Shifts in consumer sentiment and policy debates around synthetic preservatives affect short-term demand cycles, pushing manufacturers to keep improving both environmental impact and transparency. Companies like ours foresee growth in preservative blends, reduced-dose formulations, and continuous exploration of microbial resistance profiles. Process intensification, cleaner production, and full ingredient traceability will define the next wave of industry standards. Advances in analytical instrumentation allow for more precise control over batch release and impurity profiling, supporting both innovation and increased regulatory expectations. The challenge remains to match performance with consumer trust, balancing efficacy and safety with greener, more sustainable production. Persistent questions about minor metabolites and low-level chronic exposure keep us connected to external researchers and regulatory bodies. By investing in robust data and responsive process improvement, we aim to keep sodium methylparaben as a reliable, well-understood solution even as the future of preservation chemistry evolves.
What We Make — And Why People Rely on It
We’ve been manufacturing sodium methylparaben for decades, and in that time we’ve watched how this preservative earned a permanent place in both personal care and pharmaceutical sectors. This compound does one job particularly well: it extends shelf life by actively protecting products from the growth of harmful microorganisms. This practical benefit gets overlooked sometimes, but stop to think about how bottles of skin cream and oral suspensions reach bathrooms and hospital clinics months after leaving the factory. Without preservatives like sodium methylparaben, most of those items wouldn’t survive the journey, let alone stay consistently safe for use.
Formulators Trust Proven Preservation
The chemists and laboratory teams who rely on our product every year use sodium methylparaben most often to keep water-based formulas stable. We hear from customers developing everything from bubble baths to syrups for cough relief. Once water meets botanical extracts, sugars, or vitamins, microbe growth can accelerate—especially in warm or humid storage. Methylparaben’s sodium salt dissolves well in water and mixes smoothly with a wide range of raw materials. Success with batch after batch, even on industrial scale, stems from its predictability under varied conditions.
No other preservative comes with such a long track record of regulatory acceptance. The European Union, US Food and Drug Administration, and several health authorities across Asia have reviewed safety profiles and toxicology studies stretching back to the early 20th century. Our own production team keeps up with batch analysis for impurity levels to ensure compliance with relevant pharmacopeial standards. When policies shift—in concentration limits or permitted applications—we adapt our process to help customers stay up to date.
Supporting Changing Product Trends
Market tastes change. Natural and organic product launches rise every year, pushing brands to hunt for gentler preservatives or combine more than one agent to achieve broad-spectrum protection. We spend extra time with partners who want to balance minimalist “free-from” ingredient lists with standards for reliability and safety. Sodium methylparaben usually appears at low concentrations, limiting potential irritation. Its low odor and flavor make it a quieter option for dating the shelf life clock without interfering with a product’s main sensory features.
There’s no single answer for every formula challenge, but this preservative keeps proving itself worth including. One batch of sodium methylparaben added at the right stage of mixing can mean the difference between a lotion that reaches its two-year expiry with confidence and one that sours after a few weeks. Lotions, shampoos, syrups, gels, and creams often include it in their defense against spoilage.
Looking Ahead—Addressing Questions and Concerns
Public debate about synthetic additives continues in food and cosmetics. Scientific evidence and regulatory reviews draw lines for safe use, but we know trust grows through transparency. We provide technical data on purity and maintain traceability from raw material sourcing through packaging. Our door stays open to engineers, quality managers, or small makers with questions about best use in specific formulas. Sodium methylparaben’s value remains clear: it’s about keeping products safe, stable, and ready for people to use with confidence—and that’s something we’re proud to manufacture properly, every time.
Understanding What Goes into Everyday Products
From the production floor, the choices we make show up on shelves in creams, lotions, shampoos, and cleansers. Sodium methylparaben sits among the most common preservatives. Its job is to control microbes that would otherwise spoil a product in a matter of weeks. Rotting creams cause real risks, and failed preservation hurts consumer trust and safety far more than most people realize. Every manufacturing run aims for consistency. Rigid protocols confirm purity and concentration—nothing gets packed until it passes. Sodium methylparaben meets those requirements with each batch.
Safety: Evidence over Rumor
Talk around parabens can sound alarming, but fact matters more than speculation. Published data over many decades credits sodium methylparaben for a clean record when used in regulated limits. The U.S. Food and Drug Administration lists it as GRAS (generally recognized as safe) for use in food and cosmetics. In the European Union, finished products cannot exceed authorized amounts; typical cosmetic formulas use it far below this ceiling. Our plant rarely sees questions about acute allergic reactions—these show up only in rare cases, and customer feedback sees no pattern tied to our batches.
Balancing Preservation and Public Concerns
It’s true that consumer groups and some scientists question parabens’ possible links to hormonal effects. Repeated independent reviews point out that sodium methylparaben breaks down safely in the body and does not build up. The Scientific Committee on Consumer Safety re-examined these concerns and found the current allowable concentrations safe. Many personal care brands still seek “paraben-free” labeling for marketing, but without these preservatives, spoilage rates jump. Few natural alternatives can match the track record for stability and proven results. Costs rise as well: new systems need bulkier ingredients, more complex handling, or reduced shelf life. That means more waste, more returns, or higher risk of microbial contamination.
Manufacturing with Responsibility and Transparency
Responsible production starts with sourcing. We purchase raw material from thoroughly vetted partners who meet international standards for quality. Every shipment receives in-house identity testing. Once in our facility, sodium methylparaben moves under controlled conditions into tanks or mixing vessels, avoiding contamination at every stage. Material tracing backs every drum to its source and every finished batch to its records. Quality managers check finished goods by analyzing shelf-life samples over a year or more, always monitoring for degradation. Our lab retains reference samples long after the lot ships. Safety checks keep products in line with all global regulations, not just minimum requirements. Real-world incidents—a leaky shipment, tampered labels, a bad reaction—always prompt full investigation. Open communication with customers guides improvements.
Looking Ahead: Innovation Without Compromising Safety
Consumer voices drive change, and manufacturers bear responsibility to listen while standing by evidence. Demand for alternative systems increases research into botanicals, peptides, and ferment-derived options, but sodium methylparaben remains a reliable workhorse for now. As regulations evolve and new findings emerge, our production adapts. If better options deliver the same microbial control with equal or better safety, we pivot. Until then, data and vigilance—not rumor—guide every batch.
Direct Experience from the Manufacturer’s Floor
Sodium methylparaben shows up often in our production schedules. Years back, our facility shifted away from benzoic acid preservatives in some applications because customer concerns about skin sensitivity kept increasing. Sodium methylparaben stepped in with a decades-long safety record and impressive performance in controlling microbes. Every time we send a batch out the door, quality assurance tests check for impurities, residue solvents, and confirmation of active ingredient. This vigilance isn’t just for regulatory compliance. Many of our customers make personal care or pharmaceutical products, where safety and consistency shape both trust and long-term partnerships.
Allergic Reactions: Looking Past Anecdotes
Dermatologists label sodium methylparaben as a possible allergen, but allergic contact dermatitis cases trace back to a small subset of sensitive individuals. Decades of patch testing in Europe and North America yield reaction rates under 2%. Our technical teams monitor scientific literature to assess risk as chemistries evolve and consumer awareness grows. In our own follow-ups with formulation chemists and health & safety officers, clear patterns never emerge. Most flagged irritation events link to overuse or to the product’s overall formula rather than to sodium methylparaben alone.
Thousands of metric tons pass through our reactors and blending tanks each year, with the majority headed to high-volume, rinse-off products like shampoos and liquid soaps. Complaints about irritation rarely point to parabens. Instead, they stem from fragrances, colorants, or other challenging raw materials. For leave-on topical products, cautious formulators often use even lower levels than needed for antimicrobial preservation, providing an added margin of safety. Regulatory authorities continue to permit parabens such as sodium methylparaben at modest concentration limits, affirming the low observed risk in properly manufactured products.
Balancing Facts and Fear
Misinformation complicates the discussion, both for contract manufacturers like us and for global brands. Bloggers, online forums, and social media can transform isolated incidents into a narrative that colors perception. Paradoxically, the absence of broad reporting on allergic reactions sometimes gets overlooked. Among our largest clients in North America and Asia, only a handful of end-user complaints across millions of finished goods involved skin redness potentially linked to parabens, and follow-up proved inconclusive. We constantly share safety data with our partners, knowing that fear without credible evidence misdirects innovation and investment.
Real-World Solutions from Inside the Industry
For finished product makers, transparency works best. Listing sodium methylparaben on product labels in clear, unambiguous language empowers consumers with sensitive skin to make informed choices. Our technical teams help customers in the early stages of formulation to design preservative systems that couple low-risk ingredients with targeted efficacy. In dermatologically sensitive applications, we test for cumulative irritation potential on reconstructed human skin models, sharing reports with regulatory bodies and brand owners alike. Data matters more than conjecture.
Long before ingredients leave our warehouse, we practice meticulous batch tracking, documentation, and post-shipment surveys. These protocols address rare but possible allergy reports in a practical, methodical way. Where market demand shifts—such as in non-preserved or paraben-free lines—we also invest in alternatives, though these sometimes come with trade-offs in cost or effectiveness. Our job as a manufacturer is to ensure every delivery, whether it contains sodium methylparaben or not, exceeds technical and safety expectations, built on real-world experience.
As a chemical manufacturer working with parabens daily, the conversation around sodium methylparaben as a preservative comes up a lot. Sodium methylparaben is a popular ingredient in our range, used by clients in personal care, pharmaceuticals, and food industries. The main characteristic that draws formulators to it is its performance as a preservative.
How Sodium Methylparaben Defends Against Spoilage
Bacteria and fungi thrive in moist environments. Moisture attracts contamination, and without the right defensive ingredient, finished goods can lose shelf stability fast. Sodium methylparaben helps address this risk. Once blended into a product, it interrupts the growth cycle of fungi and some bacteria, mainly by targeting enzymes they rely on for normal function. Products that carry this ingredient tend to show less evidence of spoilage over time, even under typical consumer storage conditions.
The science here isn’t obscure. Methylparaben, in its base form or as sodium methylparaben, shows a proven record of inhibiting fungal growth. Pharmaceutical companies trust it to protect oral syrups and topical creams from becoming unsafe or less effective during storage. In personal care lines, the same ingredient holds back mold and yeast in creams, shampoos, lotions, and even wipes—formulas known to be prone to contamination once exposed to air or hands.
Why the Sodium Salt is Favored in Formulation
Manufacturers often switch to the sodium salt version of methylparaben because it dissolves more easily in water-based products. Water solubility helps ensure even distribution throughout large batches—important when you’re producing tens of thousands of units per day and aiming for predictable shelf life. Using sodium methylparaben also means less need for surfactants or other processing tricks to bring the preservative into solution. This reduces the chances of unwanted chemical interactions with fragrances, active ingredients, or thickeners.
Keeping Products Safe and Consumers Informed
In all the years producing this preservative, we have seen its usage undergo close scrutiny from health agencies. Regulatory reviews in Europe, North America, and Asia have set strict usage limits and required conclusive safety data. Operators need to pay careful attention to dose guidance—not just to meet legislation but also because more is not better. Excess levels may trigger skin reactions in the most sensitive consumers. Smart manufacturing keeps concentrations low while still creating a barrier against microbial problems.
There’s a loyal demand for sodium methylparaben partly because alternatives don’t always deliver the same blend of cost, ease of use, and reliable preservation. Phenoxyethanol, benzoates, or organic acids offer options for certain formulas, but each comes with its own hurdles—smell, pH sensitivity, or restricted scope. Small businesses often switch ingredients based on public perception trends, while larger players run pilot batches to track shelf-life outcomes side by side. The honest truth: without some form of reliable preservation, mold, and yeast will make themselves known every time.
Balancing Value and Responsibility
As a manufacturer, safeguarding consumer health and our clients’ reputation shapes every production decision. Quality controls, batch testing, and documentation all reflect an appreciation for scientific consensus and regulatory requirement. Sodium methylparaben continues to play a central role in the fight against spoilage, for good reason—but only as part of a broader system that respects legal limits, ingredient compatibility, and transparency with customers.
An Insider's Take from the Production Floor
Working in chemical manufacturing gives a close look at how ingredients shape products – and how regulations keep consumers safe. Sodium methylparaben stands out among the preservatives used across industries. Every so often, the question comes up: can this compound play a role in food products? Experience in production and ongoing engagement with regulatory shifts offer clarity.
Regulatory Borders Define Usage
As a producer, understanding the rulebook is the first checkpoint. Sodium methylparaben falls under the family of parabens, which gain recognition for their antimicrobial strength. In the personal care sector, demand rarely shifts, and both industry and regulators have set maximum use levels. In the food sector, rules become stricter.
Looking at the global landscape, agencies like the United States Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA) keep parabens such as methylparaben, ethylparaben, and propylparaben under constant review. In the United States, methylparaben and propylparaben see limited use in food, with federal law requiring detailed labeling and strict concentration caps. China also allows specific parabens in certain foods, but sodium methylparaben doesn’t appear on approved lists for food use in most developed markets. That absence speaks volumes. National standards, such as the European Union’s food additive regulations, restrict sodium methylparaben outright for food applications. These decisions rest on toxicological studies examining potential health concerns over cumulative intake and possible endocrine disruption. It's not just paperwork — these directives shape every order received and every batch that leaves our facility.
Safety and Trust Must Guide the Production Line
Parabens can act as efficient preservatives in water-based systems thanks to their broad-spectrum activity against molds and bacteria. In cosmetics and pharmaceuticals, our customers count on this. But for foods, public trust hinges on strict adherence to the approved additives. It doesn’t matter if sodium methylparaben has a similar safety profile to other parabens at low exposure levels. Companies manufacturing additives steer clear of supplying products where food use doesn't align with established regulatory lists.
From daily operations, there’s constant pressure to meet demand for clean label solutions. The market often asks about substitutes as manufacturers phase out certain preservatives. Efforts focus on naturally derived alternatives or other synthetics cleared by regulators. New preservatives receive scrutiny, demanding lengthy review before anyone can commercialize them for food. Research and pilot scale investment goes into alternatives like potassium sorbate, sodium benzoate, or natamycin, all of which have GRAS or E-number statuses for many food categories.
Accountability Goes Beyond Compliance
The push to innovate doesn’t mean taking risks with food safety. If anything, it means upholding discipline around what goes into formulations destined for the dinner table. Inquiries about sodium methylparaben’s compatibility with food keep coming up – and manufacturers must stay transparent, lean on established science, and refuse shortcuts. In reality, sodium methylparaben’s place remains with non-food products, regardless of how effective it could be as a preservative.
Transparency, strict regulatory adherence, and open dialogue with customers create the backbone of how manufacturers maintain trust. By recognizing the limits and the science behind them, the industry keeps food safe and consumers confident.