Sodium Propyl Paraben: A Perspective from the Chemical Manufacturer’s Floor
Tracing the Past: The Roots of Sodium Propyl Paraben
Sodium propyl paraben’s journey began in the early chapters of preservative chemistry, born out of the growing demand for alternatives to benzyl-based preservatives. Chemists explored alkyl paraben derivatives, and propyl paraben surfaced as a promising candidate. The sodium salt emerged soon after for its water solubility, making it fit for many aqueous systems. Legacy chemical manufacturers, who watched formulas shift through decades, recall formulators transitioning to sodium salts in the mid-20th century, targeting food, pharmaceutical, and personal care products that called for broad-spectrum protection. Today’s familiarity with sodium propyl paraben can be traced to these pragmatic modifications sparked by practical process requirements and regulatory considerations.
Understanding Sodium Propyl Paraben: The Product’s Core
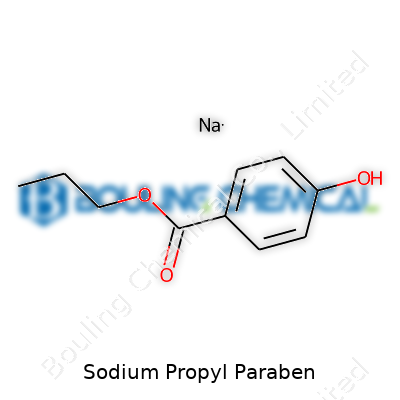
As manufacturers, experience with sodium propyl paraben starts with bulk white crystalline powder—sodium 4-hydroxy-2-propylbenzoate by IUPAC nomenclature. Odor remains faint and sweet, and the substance dissolves easily in water. This physical profile lets it blend directly into water-based product matrices without additional solubilizers. Consistent melting points and reliable particle size reflect strict process control, which matters because product variability undermines both preservation and regulatory audits. Our lab teams monitor these properties batch-to-batch to maintain transparent quality, because end users from food to pharma require predictable performance, not surprise reactivity or off-odors.
The Chemical Itself: Hard Facts and Specifications
Sodium propyl paraben sits at the molecular formula C10H11NaO3. The white granules or powder often carry a defined purity—usually not less than 98 percent. Moisture content stays under 5 percent, and residual solvents must be below threshold set by current pharmacopeias. Precise labeling details, such as batch number and storage advice, remain a fixture for regulatory and recall readiness. During internal audits, attention goes beyond the product; container integrity, traceable raw materials, and correct hazard labeling all receive scrutiny. The point isn’t just to meet regulations, but to provide assurance to any buyer downstream that no steps were skipped or corners cut.
Making Sodium Propyl Paraben: Inside the Factory
Most sodium propyl paraben starts with an esterification of p-hydroxybenzoic acid and n-propanol under acid catalysis, forming the propyl paraben ester. Alkaline saponification then splits the ester, with sodium hydroxide neutralizing the free acid, producing the sodium salt. Each manufacturer tailors conditions—temperature, pressure, solvent choice—for optimal yield, energy use, and minimal impurity carryover. Experienced plant managers know that solvent residues, incomplete reactions, and trace metals must get purged in downstream crystallizations and washes. Wastewater and solvent streams run through closed-loop treatments to keep environmental load low. Operators follow standard operating procedures rigorously, as deviations lead to off-quality and risk batch discards.
Chemical Reactions and Derivative Work
We’ve seen interest from R&D labs and custom chemical teams to tweak the core structure of sodium propyl paraben. Efforts to extend chain lengths or introduce substituents bring shifts in solubility, preservative spectrum, and allergenicity. Laboratories sometimes explore partial esterification or co-crystallization with other parabens to broaden antimicrobial protection or fit niche solvent systems. Yet, any attempt to change the base molecule must weigh regulatory classification and toxicological testing. Reaction conditions—strong bases, acids, or heat—can decompose or modify the paraben, yielding side products that require careful analysis before a new grade can progress. We invest in both in-house and third-party assessment to make clear what’s produced before offering a novel variant.
Names and Identifiers: Speaking the Language
In day-to-day conversation among formulation chemists and purchasing managers, sodium propyl paraben goes by several names: sodium propyl p-hydroxybenzoate, sodium 4-hydroxybenzoate propyl, or simply E217 in food compliance systems. The CAS number offers a permanent reference, cutting through linguistic ambiguity on global shipments. International standards—JP, EP, USP—each list nomenclature and grade guidelines, crucial for pharmaceutical or food customers who audit against these references. Internally, SAP codes and inventory labels align with these identifiers for swift logistics and recall precision.
Safety, Handling, and Regulatory Duty
Every facility handling sodium propyl paraben must stay vigilant on worker safety and environmental stewardship. Powdered product requires dust control—extraction, filtration, and protective clothing—because inhalation or eye contact brings discomfort. Our teams undergo regular training on safe handling, response protocols, and waste segregation. While sodium propyl paraben rates low on acute toxicity scales, manufacturers watch for cumulative handling, storage near oxidizers, and accidental release. Regulatory frameworks—REACH in Europe, EPA in the USA, CFDA China—shape permissible exposure limits, labeling, and registration. Up-to-date Safety Data Sheets stay accessible on site and externally for transparency.
Where Sodium Propyl Paraben Works
End users choose sodium propyl paraben for its proven inhibition of yeasts and molds in a range of product types. Manufacturers routinely supply it to personal care, food, and pharmaceutical sectors, where liquid creams, gels, and oral suspensions benefit from its high water solubility and compatibility with other preservative blends. Food packers rely on E217 in processed meats, bakery goods, and confectionery to extend shelf life. Healthcare product formulators turn to it for cough syrups, suspensions, and dermatologic creams, where clarity and consistent dose matter. Success depends on accurate dosing—too little sacrifices microbial protection, too much may trigger regulatory breach.
Continuous Investment: The Role of R&D
Laboratories at producing facilities continually search for new uses and process improvements. Teams develop rapid-dissolving grades, micronized powders, or grades with ultraviolet-protection packaging to forestall degradation. Collaborations with academic groups or industry consortia help anticipate shifts in microbial resistance and consumer sensitivities. Simulation testing, challenge studies with fungi and bacteria, and shelf-life validation drive ongoing improvements. These projects often uncover new compatibilities or challenge old dosage conventions, reflecting real-world learning, not just literature review.
Unpacking Toxicity Research
Long-term studies shaped regulatory stances on parabens, and sodium propyl paraben received intense scrutiny. Animal and human models confirm low acute oral and dermal toxicity at permitted use levels. Debate around potential endocrine-disrupting effects prompted extensive studies. Most respected health agencies, including the US FDA and European Food Safety Authority, continue to support sodium propyl paraben’s use within established limits, citing comprehensive reviews. That said, vigilance continues. Every batch gets tested for trace contaminants like residual solvents and heavy metals, keeping confidence high both for customers and end-use regulators.
Looking Forward: The Future of Sodium Propyl Paraben
Shifts in consumer preference and global regulation signal challenges and opportunities for sodium propyl paraben producers. Some markets push toward “preservative-free” products, but microbial stability cannot be ignored. Innovations now target even cleaner synthesis routes, higher-purity grades, and blends with other natural or low-impact preservatives. Advanced analytics monitor for ever-lower contaminant thresholds, and digital quality records support the traceability modern buyers demand. Sustainability, both in production and packaging, shapes every improvement project. Close work with downstream customers and regulators will keep sodium propyl paraben relevant as long as product safety, shelf life, and cost matters to manufacturers and consumers alike.
Understanding Where Sodium Propyl Paraben Makes a Difference
Every day, behind the labels of lotions, shampoos, ointments, and syrups, factories run with precision. As a chemical manufacturer, we follow closely how each compound behaves in different settings. Sodium propyl paraben has carved out its spot thanks to its reliability. Preserving finished goods—keeping them free of mold, yeast, and harmful bacteria—takes genuine expertise. Using this preservative helps extend shelf life and protect consumers at the same time.
This substance appears most often in the personal care sector. Think of those familiar creams stacked on supermarket shelves. Each batch faces a risk of microbial contamination, especially since water or botanical extracts feature strongly in modern formulas. Applying sodium propyl paraben gives these products a fighting chance. With the proper dosage, creams keep texture, appearance, and safety longer. Testing shows that creams left unsecured lose their freshness much faster. Responsible manufacturers use preservatives measured to guidelines so the final product not only passes quality checks but arrives on store shelves with sufficient protection until opened by the customer.
Formulators who design shampoos and liquid cleansers rely on its broad compatibility. The salt form (sodium propyl paraben) dissolves smoothly in water, making it a practical option for liquid blends. Using this ingredient simplifies the process because formulators know its solubility matches most rinse-off and leave-on applications. To put it plainly, this compound works well with both oily and watery mixes. Instead of switching strategies for each product, a manufacturer can adopt a consistent approach, saving time and reducing the likelihood of failed batches.
Beyond the world of shampoo and lotion sits the realm of pharmaceuticals. Calibrating shelf life and minimizing spoilage in syrups or topical medicines means more efficiency for logistics and less concern about wasted inventory. Here, sodium propyl paraben helps punch back against microbes, reducing the risk of spoilage before these products reach a pharmacy. Proper preservation aligns with medicine quality and consumer trust because people expect the same standard whether the bottle spends days in a warehouse or months in a bathroom cabinet.
Not every challenge gets solved just by adding a preservative. Regulatory bodies—such as the US FDA and European authorities—regularly review safety data. Science changes, and customer preferences shift. As a manufacturer, we track these updates, adjust usage rates, and pay close attention to purity. Overuse complicates formulations, and underuse gives microbes room to grow. Precise measurements matter. All production runs undergo microbiological testing so that the trade-off between product safety and ingredient minimalism lands in the right place.
Every decision in our plant links back to real-world use. Some brands now pursue alternatives for certain product lines based on consumer expectations for “preservative-free” claims. Still, where risks from contamination remain high, sodium propyl paraben stays on the bench for one reason: proven, dependable defense against unwanted growth. Years of laboratory and production experience tell us which mixes demand extra care and which can get by with less.
Keeping pace with changing scientific consensus and market trends sits at the core of our commitment. Only by listening to feedback—from technical teams, labs, regulators, and end-users—do we improve. The humble molecule behind so many safe, long-lasting bottles on store shelves represents more than chemistry; it reflects a legacy of careful manufacturing designed to keep real people safe.
Real-World Thoughts from a Chemical Manufacturer
The conversation about sodium propyl paraben in cosmetics tends to get complicated. As a manufacturer, every raw material that passes through our production lines gets scrutinized. It’s not enough to simply stamp and ship—our reputation and the trust of the end user ride on every decision. Over the years, this class of preservatives has drawn more than its fair share of attention. So, let’s talk about our experience and why sodium propyl paraben keeps surfacing in cosmetic production debates.
Parabens in general have a long history in personal care products. They stop spoilage and keep both brand-new and half-used lotions and creams from breeding harmful microbes. Sodium propyl paraben stands out for its stability and broad-spectrum preservation, good solubility, and mildness. These properties help formulators stabilize creams without needing harsh alternatives or preservatives that trigger allergic reactions or skin sensitivity in broader groups.
Safety still comes up a lot, and for good reason. The scientific consensus points to low concentrations of sodium propyl paraben in cosmetics as safe for consumers. Extensive studies, along with reviews from recognized authorities like the US Food and Drug Administration and the European Commission’s Scientific Committee on Consumer Safety, have examined data from decades of use. Researchers have looked for hormone disruption at everyday exposure levels and have not found effects in line with public concerns. In the laboratory, occasional incidents of mild skin irritation appear at much higher concentrations than what goes into shelf-ready products.
Strict regulatory controls keep this conversation on track. European rules, for example, limit use to concentrations judged by scientific review as non-threatening, with safety margins built in. Our quality control labs check every batch, verifying that finished products stay far below those legal limits. The process isn’t negotiable. Batches that don’t pass get destroyed.
Some pressure comes from customer questions. Retailers and brand-owners knock on our door with requests for paraben-free options, often fueled by social media or changing consumer trends. We talk through the options, including potential issues if preservatives get tossed out without replacements. Yeast, bacteria, and mold don’t care about public debate—they grow wherever they find holes in the defense. In those cases, shelf life drops, spoilage spikes, and sometimes more irritating or less studied chemicals push their way in. Anyone buying “preservative-free” should look closely at production and expiry dates, and at what else replaces traditional preservatives.
Manufacturers take quality and safety seriously because nobody wins if a customer gets hurt. Raw material traceability, regular audits, and independent third-party tests give us a strong grip on what leaves our site. Environmental responsibility also factors in—paraben breakdown rates in nature and wastewater treatment inform our policies. We monitor new research and jump when regulations tighten because no supplier wants recalls or recalls’ fallout.
With decades of data and strict controls, sodium propyl paraben remains a sound choice for preserving cosmetic and skincare products. Ongoing research and feedback from actual users keep the process honest. If future investigations uncover new facts, the manufacturing world adapts. Until then, every batch reflects careful decision-making rooted in science, practical experience, and the real-world safety of consumers and workers alike.
Industry Perspective from the Manufacturing Floor
Our factory runs nearly nonstop, producing a variety of preservatives that allow everyday products to reach homes and businesses safely and reliably. Sodium propyl paraben stands among the preservatives that have supported food, pharmaceutical, and personal care markets for decades. Through years of daily handling and large-batch production, we’ve seen and heard just about every concern customers and partners can bring up about potential health effects.
Looking Closer at Sodium Propyl Paraben
Sodium propyl paraben lands on the “safe list” for most health authorities worldwide. It doesn’t stick around in human tissue, and workers in our plant have regular health checks without a pattern of problems linked to this compound. Structurally, the molecule belongs to the paraben family—esters of para-hydroxybenzoic acid. These get added in tiny amounts to act against mold and bacteria, addressing both safety and shelf life.
Like all parabens, this compound gets talked about for possible allergy triggers. Most of our team rarely sees true allergic reactions. Medical literature backs this up—documented cases exist, but numbers remain substantially lower than those linked to fragrances, dyes, or other common additives. Some of our customers have run their own patch tests and find that irritant responses triggered by sodium propyl paraben often become visible only with higher-than-normal exposure, sometimes involving already damaged or fragile skin.
What Triggers Issues?
For most healthy users, side effects rarely crop up. In our experience, reactions more often happen to people who have a history of multiple allergies or underlying skin conditions. Signs described by clinics include mild redness, itching, or swelling—usually after direct and repeated contact. Rarer, but possible, are hives or rashes that come up as immune system alerts. In our lines, we follow dust control and hygiene protocols, and incident reports remain quite rare. The real risks surface mostly in niche settings, such as individuals who already react to many ingredients.
Why Does Reassurance Matter?
Science and safety guidelines direct how much goes into finished goods—not just our own, but across the industry. Most countries cap the allowable percentage far below the levels that promote irritation. Over the years, we’ve tweaked processes to guarantee consistent, traceable output, letting our buyers respond confidently if a customer voices a concern about potential side effects. Few additives match the decades-long record and scientific support behind this preservative group.
Misinformation sometimes circulates about parabens causing hormone disruption or cancer. High-quality independent studies run over many years have not found solid links at realistic, permitted usage levels. We monitor the ongoing research, and regulations have shifted in some regions, but this comes from a mindset of caution rather than red flags raised by real-world cases.
Solutions for Sensitive Customers
For clients serving highly sensitive or allergy-prone populations, alternate preservative strategies exist. Our technical teams have formulated blends that omit parabens entirely or switch to different chemistries. Whenever customers request it, we supply batch-specific information and support their risk assessment, since traceability counts for both ethical and regulatory standards. Communication between manufacturers, brands, and buyers ensures peace of mind for everyone along the supply chain.
Every customer places trust in our oversight. We keep working with health professionals, regulators, and researchers to update our practices. Questions about sodium propyl paraben crop up year after year, but for the vast majority, this ingredient delivers time-tested safety at scale. For those rare situations where allergy risk exists, it pays to listen, document, and tailor solutions—not just because the regulations say so, but because that’s how responsible manufacturing survives and serves future generations.
Regulators and Real-World Experience with Sodium Propyl Paraben
Working as a manufacturer, the question of regulatory status for different chemicals is one I face every day—especially with preservatives like sodium propyl paraben. Years ago, it would show up on most order requests for personal care and pharmaceutical products. Expectations around regulatory approval have changed a lot, and this is a good place to talk openly about what’s really happening with sodium propyl paraben and why buyers keep asking about its approval.
Sodium propyl paraben falls in the paraben group: synthetically made, valued for strong antimicrobial action, and easy to formulate for water-based systems. Our chemists still recognize its stability, but use in finished products is shaped almost entirely by regulatory trends, scientific study, and public perception. We track these factors closely, since confidence in any chemical depends on how regulators view its risk profile and how laws shift from region to region.
Levels of Approval: Largely Restricted for Human Use
The strictest rules appear in the EU market, where regulatory authorities ban the use of sodium propyl paraben in cosmetics and personal care under Commission Regulation (EU) No 358/2014. In daily manufacturing, this ban directly means removing this paraben from any formula destined for European shelves. We’ve spent years reformulating and searching for alternatives because the ban limits not just what companies can sell, but also how we plan batch production and stock raw materials.
The United States FDA lists parabens like methylparaben and propylparaben as generally recognized as safe (GRAS) for limited food and pharmaceutical uses at specified concentrations. But sodium propyl paraben does not feature on the FDA’s approved lists for addition to food, and FDA’s labeling requirements force brands to rethink transparency when using any paraben. Since most queries come from international clients, navigating these patchwork rules takes up time and resources, especially since every order can be audited by customs or port officials. In practice, major global manufacturers have phased out sodium propyl paraben in favor of ingredients that won’t cause delays or detainment in ports.
Data Behind the Restrictions
Decisions to pull sodium propyl paraben from allowed ingredient lists stem from concerns over possible endocrine-disrupting activity. Health authorities review animal data and exposure studies, then act on the side of caution when results indicate disruption, even at low use levels. I’ve seen this play out repeatedly: once one major market restricts a chemical, global buyers cut usage preemptively to avoid market losses. In the case of sodium propyl paraben, enough scientific concern—combined with consumer wariness—creates a domino effect, and that shapes the whole supply chain. As manufacturers, we stay proactive about sourcing and testing new preservative systems that pass toxicological reviews.
Working Towards Solutions
We have learned the importance of transparency. Any batch we develop comes with up-to-date documentation on regulatory status—sourced right from the current registers. If regulations evolve, we adjust fast. Sourcing alternatives such as sodium benzoate or potassium sorbate, which have wider international acceptance, means we help customers avoid disruption. Our product development now involves direct collaboration with regulatory experts to anticipate bans before they happen, saving both compliance headaches and wasted inventory. In the end, the regulatory status of sodium propyl paraben serves as a real-world reminder of how rules, science, and consumer trust drive product choices from the factory floor to the retail shelf.
Introduction
Daily, we answer questions about ingredient safety from professionals and brands developing skincare products for sensitive skin. Sodium propyl paraben, a preservative used in personal care and pharmaceutical formulas, often comes up in these conversations. Years of experience in manufacturing this compound have shown us why opinions can differ, but also revealed some guiding facts about its use with delicate skin types.
Understanding Sodium Propyl Paraben’s Role
Preservatives like sodium propyl paraben play a crucial role by defending creams, lotions, and other water-based products against bacteria, mold, and yeast growth. In low concentrations, sodium propyl paraben efficiently extends product shelf life without an overwhelming load of chemical additives. Many global health authorities, including the European Commission, have reviewed this class of preservatives, confirming their safety at recommended limits.
Skin Sensitivity and Real-World Experience
Sensitive skin reacts more readily to contact allergens and harsh substances. Through decades of real-world feedback from brand formulators, dermatologists, and our own internal safety tests, most individuals, even those with sensitive skin, do not react to sodium propyl paraben when used within 0.1% to 0.3%. Reports of adverse skin reactions to this ingredient remain rare compared to certain fragrance compounds, essential oils, or less-refined natural preservatives.
Occasionally, someone experiences mild irritation due to previous sensitization or a skincare routine already under stress from multiple actives or environmental factors. No single ingredient fits every person, but good formulation practices limit these risks. Parabens, including sodium propyl paraben, do not increase photosensitivity or thin the skin barrier, unlike potent actives such as retinoids or exfoliating acids.
Debates and Misinformation
Several years ago, concerns circulated in popular media about paraben safety, sparking anxiety among consumers. Many large brands reformulated products or swapped them for less-studied alternatives. Yet, as manufacturers, we monitor peer-reviewed studies and regulatory reviews closely. Consistent data shows sodium propyl paraben remains stable, non-reactive, and does not bioaccumulate. Allergic skin reactions occur in an extremely small fraction of individuals, and patch tests with controlled concentrations support this conclusion.
Natural preservatives often sound appealing, but repeated feedback highlights that these alternatives can bring more risk of allergies, shorter shelf life, or unpredictable breakdown in complex formulas. Sodium propyl paraben allows brands to keep products stable with lower preservative amounts, limiting total exposure for end-users.
Best Practices and Solutions
Sensitive skin formulas thrive with minimal fragrance, limited active ingredients, and gentle preservatives. Using sodium propyl paraben at carefully controlled doses fits these requirements. Finished product testing—repeated both in-house and externally—remains the most practical way to catch sensitivities before product launch. Open dialogue with dermatologists and consumer panels also helps match ingredient safety with real user experience.
Manufacturers achieving low-irritation claims often pair sodium propyl paraben with skin-loving bases and a simple formula lineup. Encouraging brands to publish transparency about ingredients and their concentrations builds trust, letting end-users make informed choices suited to their own skin needs.
Conclusion
Every ingredient comes under scrutiny as trends in skin health evolve, but sustained evidence and daily production experience support the careful use of sodium propyl paraben in sensitive skin care. Responsible formulation, clear communication, and rigorous testing continue to give consumers the safest possible options.